Aldol
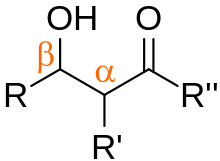
In
Aldols are the product of a carbon-carbon bond-formation reaction, giving them wide applicability as a pre-cursor for a variety of other compounds.
Synthesis and reactions
Aldols are usually synthesized from an
Aldols may also undergo a condensation reaction in which the hydroxy group is replaced by a pi bond. The final structure is a reactive α,β-unsaturated carbonyl compound, which can also used in a variety of other reactions:
- RC(O)CH2CH(OH)R' → RC(O)CH=CHR' + H2O
Applications
Aldols synthesized from two aldehydes are usually unstable, often producing secondary compounds such as diols, unsaturated aldehydes, or alcohols.[1] Hydroxypivaldehyde is a rare example of a distillable aldol.[3] The aldol 3-hydroxybutanal is a precursor to quinaldine, which is a precursor to the dye quinoline Yellow SS.[1]
Aldols are also used as intermediates in the synthesis of polyketide natural products and drugs such as Oseltamivir and Epothilone.[4][5][6][7]
See also
References
- ^ ISBN 9783527303854. Retrieved 1 April 2023 – via Wiley Online Library.
- ^ PubChem. "CID 21282929". National Center for Biotechnology Information. Retrieved 2023-04-15.
- – via ACS Publications.
- PMID 17103481.
- PMID 30443084– via Thieme.
- PMID 20866058– via National Library Of Medicine.
- ISBN 978-3-527-30714-2, retrieved 2024-04-15
