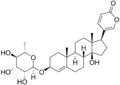
Arenobufagin

| |
| Names | |
|---|---|
| IUPAC name
5-[(3S,5R,10S,11S,13R,14S,17R)-3,11,14-trihydroxy-10,13-dimethyl-12-oxo-2,3,4,5,6,7,8,9,11,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]pyran-2-one
| |
| Other names
Arenobufagin
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C24H32O6 | |
| Molar mass | 416.514 g·mol−1 |
| Appearance | liquid |
| Density | 1.4±0.1 g/cm3 |
| Boiling point | 637.2±55.0 °C at 760 mmHg |
| Vapor pressure | 0.0±4.3 mmHg at 25 °C |
Refractive index (nD)
|
1.622 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Cardiotoxic |
| GHS labelling: | |

| |
| Danger | |
| Flash point | 219.3±25.0 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Arenobufagin is a
Sources
The compound arenobufagin is one of the major components of
History
Arenobufagin is a component of a certain toad venom which goes by the name of Chan’su. For centuries, this venom has been used in
Function in medicine
As already mentioned, arenobufagin has been widely used in traditional Chinese medicine (TCM) to tackle down carcinogenesis , since it inhibits cell growth in several cancer cells. It is one of the central active ingredients of toad venom for treatment.[5] This happens both in purified form or in combination with other ingredients, which are mostly herbal components. Toad venom is in fact still used in clinical practice of in TCM to treat hepatocellular carcinoma (HCC).[6] So far there is still little known about the anti-angiogenic properties of Arenobufagin. One studie shows that Arenobufagin inhibits VEGF-induced endothial cell tube formation.[7]
Intake
Under normal circumstances, arenobufagin is a
Toxicodynamics

Arenobufagin is believed to play a role in the regulation of the transport of water and electrolytes across
Besides its effects on the Na+-K+ pump, arenobufagin also has some other effects on
Detoxification
The biotransformation of arenobufagin by Alternaria alternata leads to the following three metabolites: 3-oxo-arenobufagin (1a), ψ-bufarenogin (1b),[9] and 3- oxo- ψ-bufarenogin (1c). The biotransformation processes consists of a main reaction whereas the dehydrogenation of the 3-hydroxyl group takes place. This process is followed by isomerization.[10]
Similar forms
Arenobufagin is a derivative of the so-called bufadienolides, where it also has a steroid structure. A characteristic of bufadienolides is that they all contain two double bonds in the lactone ring.
Symptoms
Circulatory system
Arenobufagin works like Cardiac glycosides. It inhibits the
Digestive system
Arenobufagin has shown to cause appoptose in hepatocellular carcinoma cells in mice, although this method is not used to cure hepatocellular carcinoma in modern human medicine.
Structure-activity relationships
There is a rather large homology in structure between arenobufagin and
-
Structure of arenobufagin
-
Structure of a cardiac glycoside, proscillaridin
Toxicity
Acute toxicity
Although a low dosis of arenobufagin can be used as a medicine of heart rate problems, a high dose can lead to acute heart problems and even death. Arenobufagin is also toxic for hepatocellular carcinoma cells, which is a positive result for the body.
Chronic exposure
Toxicity because of chronic exposure was not clearly documented thus far. However it is discussed that the chronic exposure to this compound can cause the development of
See also
- Bufagin
- Bufadienolide
- Cinobufagin
- Gamabufagin
- Marinobufagin
- Quercicobufagin
- Regularobufagin
- Vallicepobufagin
- Viridibufagin
- Digitalis
References
- PMID 3127947
- PMID 8223897
- PMID 22305746.
- ^ Zhang, D. M., et al. (2013). "Arenobufagin, a natural bufadienolide from toad venom, induces apoptosis and autophagy in human hepatocellular carcinoma cells through inhibition of PI3K/Akt/mTOR pathway." Carcinogenesis 34(6): 1331-1342.
- ^ Li M, Wu S, Liu Z, Zhang W, Xu J, Wang Y, Liu J, Zhang D, Tian H, Li Y, Ye W. (2012). “Arenobufagin, a bufadienolide compound from toad venom, inhibits VEGFmediated angiogenesis through suppression of VEGFR-2 signaling pathway.” Biochemical Pharmacology. 83, 1251–1260
- ^ Tang,J. et al. (2008) “Research progress on clinical application of venenum bufonis preparation as antineoplastic drug.” China Pharmaceuticals, 17, 15-16.
- ^ Li M, Wu S, Liu Z, Zhang W, Xu J, Wang Y, Liu J, Zhang D, Tian H, Li Y, Ye W. (2012). “Arenobufagin, a bufadienolide compound from toad venom, inhibits VEGFmediated angiogenesis through suppression of VEGFR-2 signaling pathway.” Biochemical Pharmacology. 83, 1251–1260
- ^ Zhang, D. M., et al. (2013). "Arenobufagin, a natural bufadienolide from toad venom, induces apoptosis and autophagy in human hepatocellular carcinoma cells through inhibition of PI3K/Akt/mTOR pathway." Carcinogenesis 34(6): 1331-1342.
- PMID 20432230.
- S2CID 84624677.