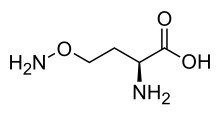
Canaline
| |
| Names | |
|---|---|
| IUPAC name
(2S)-2-Amino-4-aminooxy-butanoic acid
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10N2O3 | |
| Molar mass | 134.135 g·mol−1 |
| Density | 1.298 g/mL |
| Melting point | 213 °C |
| Boiling point | 378.1 °C (712.6 °F; 651.2 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
This article relies largely or entirely on a single source. (February 2015) |
l-Canaline (
legumes that contain canavanine, from which it is produced by the action of arginase. The most common-used source for this amino acid is the jack bean, Canavalia ensiformis
.
Toxicity
l-Canaline is the only naturally occurring amino acid known that has an O-alkyl
Tobacco hornworm
larvae fed a diet containing 2.5 mM canaline showed massive developmental aberrations, and most larvae so treated died at the pupal stage. It also exhibits potent neurotoxic effects in the moth.

Its toxicity stems primarily from the fact that it readily forms
oximes with keto acids and aldehydes, especially the pyridoxal phosphate cofactor of many vitamin B6-dependent enzymes. It inhibits ornithine aminotransferase
at concentrations as low as 10 nM.
Plant nutrition
l-Canaline is a substrate for
argininosuccinic acid synthetase. l-Canavaninosuccinic acid is cleaved to form l-canavanine by argininosuccinic acid synthetase. By these sequential reactions, the canaline-urea cycle (analogous to the ornithine-urea cycle) is formed. Every time a canavanine molecule runs through the canaline-urea cycle, the two terminal nitrogen atoms are released as urea. Urea is an important by-product of this reaction sequence because it makes ammonia (urease-mediated) that is available to support intermediary nitrogen metabolism. l-Canaline can be reductively cleaved to l-homoserine
, a non-protein amino acid of great importance in the formation of a host of essential amino acids. In this way, the third nitrogen atom of canavanine enters into the reactions of nitrogen metabolism of the plant. As homoserine, its carbon skeleton also finds an important use.
References
- Rosenthal, Gerald A. (1982). Plant non-protein amino and imino acids: biological, biochemical, and toxicological properties. Boston: Academic Press. ISBN 0-12-597780-8.[page needed]
