Cryptand
In
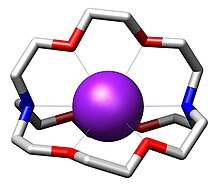
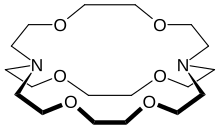
Structure
The most common and most important cryptand is N[CH2CH2OCH2CH2OCH2CH2]3N; the systematic
Properties
Cation binding
The three-dimensional interior cavity of a cryptand provides
Anion binding
Polyamine-based cryptands can be converted to polyammonium cages, which exhibit high affinities for anions. [8]
Laboratory uses
Cryptands enjoy some commercial applications (e.g. in homogenous-time-resolved-fluorescence, HTRF, technologies using Eu3+ as central ion). More importantly, they are reagents for the synthesis of inorganic and organometallic salts. Although more expensive and more difficult to prepare than crown ethers, cryptands bind alkali metals more strongly.[9] They are especially used to isolate salts of highly basic anions.[10] They convert solvated alkali metal cations into lipophilic cations, thereby conferring solubility in organic solvents to the resulting salts.
Referring to achievements that have been recognized in textbooks, cryptands enabled the synthesis of the
9.[13]
Although rarely practical, cryptands can serve as
See also
- Clathrate
- Clathrochelate
- Cryptophane
- Cyclodextrin
References
- PMID 11457025.
- ISBN 0-471-95057-2.
- ^ Lehn, J. M. (1995). Supramolecular Chemistry: Concepts and Perspectives. Weinheim: VCH.
- PMID 25138490.
- ^ 23978-09-8
- .
- .
- S2CID 22334242.
- .
- ISBN 0-08-040610-6.
- S2CID 93768664.
- ISBN 0-12-352651-5
- .
- .
General reading
- Lee, J.D. (1991). Concise Inorganic Chemistry (4th ed.). New York: Chapman & Hall. pp. 306–308 & 353. ISBN 0-412-40290-4.
- Krakowiak, K. E.; Bradshaw, J. S.; An, H.-Y.; Izatt, R. M. (1993). "Simple methods for the preparation of cryptands". .
