Polyamine
A polyamine is an
Natural polyamines
Low-molecular-weight linear polyamines are found in all forms of life. The principal examples are the triamine spermidine and the tetraamine spermine. They are structurally and biosynthetically related to the diamines putrescine and cadaverine. Polyamine metabolism is regulated by the activity of the enzyme ornithine decarboxylase (ODC).[2] Polyamines are found in high concentrations in the mammalian brain.[3]
- Natural polyamines
Synthetic polyamines
Ethyleneamines are a commercially-important class of synthetic polyamines with ethylene (−CH2CH2− linkages); global production capacity was estimated at 385,000 tonnes in 2001.[4] They are chemical intermediates often used to make surfactants and as crosslinkers for epoxy resins.[5] Some interesting members of this class include:
- EDTA (ethylenediaminetetraacetic acid). Permethylated, ethylenediamine yields tetramethylethylenediamine (TMEDA) that has a very high affinity for lithium ions.[6]
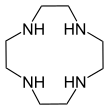
- crown ethers: 1,4,7-triazacyclononane ((NHCH2CH2)3) and cyclen ((NHCH2CH2)4). A related tetraaza macrocycle is cyclam.
- polyethyleneamine process. A related tripodal polyamine is 1,1,1-tris(aminomethyl)ethane. These are interesting chelating ligands.
- Polyethylenimine is a polymer derived from aziridine.
Other synthetic polyamines include
- Synthetic polyamines
-
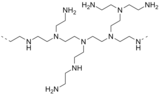
Subunit of polyethylenimine
-
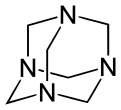
Hexamethylenetetramine with its adamantane-type structure
Biological function
Although it is known that the biosynthesis of polyamines is highly regulated, the biological function of polyamines is only partly understood. In their cationic ammonium form, they bind to
or Ca2+
, which are point charges). They have also been found to act as promoters of programmed ribosomal frameshifting during translation.[8]
Inhibition of polyamine biosynthesis retards or stops cell growth. The provision of exogenous polyamines restores the growth of these cells. Most eukaryotic cells express a polyamine-transporting ATPase on their cell membrane that facilitates the internalization of exogenous polyamines. This system is highly active in rapidly proliferating cells and is the target of some chemotherapeutics currently under development.[9]
Polyamines are also modulators of a variety of ion channels, including NMDA receptors and AMPA receptors. They block inward-rectifier potassium channels so that the currents of the channels are inwardly rectified, thereby the cellular energy, i.e. K+
ion gradient across the cell membrane, is conserved. In addition, polyamine participate in initiating the expression of SOS response of Colicin E7 operon and down-regulate proteins that are essential for colicin E7 uptake, thus conferring a survival advantage on colicin-producing E. coli under stress conditions.[10]
Polyamines can enhance the permeability of the blood–brain barrier.[11]
They are involved in modulating senescence of organs in plants and are therefore considered as a plant hormone.[12] In addition, they are directly involved in regulation of programmed cell death.[13]
Homology-directed DNA repair
Polyamines promote homologous recombination (HR)-mediated double-strand break (DSB) repair.[14] Polyamines enhance the DNA strand exchange activity of RAD51 recombinase. Depletion of polyamines sensitizes cells to genotoxic substances such as ionizing radiation and ultraviolet radiation. The effect of polyamines on RAD51 arises from their ability to enhance the capture of homologous duplex DNA and promote RAD-51-mediated homologous DNA pairing and exchange activity.[14] Polyamines appear to have an evolutionarily conserved role in regulating recombinase activity.
Biosynthesis of spermidine, spermine, thermospermine

Spermine is synthesized from the reaction of spermidine with SAM in the presence of the enzyme spermine synthase.
The polyamines undergo rapid interconversion in the polyamine cycle, in which putrescine leads to synthesis of spermidine and spermine, with degradation of these polyamines to form putrescine, which can begin the cycle again.[15]
Thermospermine (NH2−(CH2)3−NH−(CH2)3−NH−(CH2)4−NH2) is a structural isomer of spermine and a novel type of plant growth regulator. It is produced from spermidine by the action of thermospermine synthase, which is encoded by a gene named ACAULIS5 (ACL5).[16]
Polyamine analogues
The critical role of polyamines in cell growth has led to the development of a number of agents that interfere with polyamine metabolism. These agents are used in
See also
References
- ISBN 978-3527306732.
- S2CID 21063248.
- ISBN 978-1-4757-0614-7.
- ISBN 9780471238966.
- ISBN 978-0-521-78284-5.
- ISBN 0-471-93623-5.
- .
- PMID 21303766.
- PMID 14613316.
- PMID 16549429.
- PMID 19236044.
- S2CID 21925829.
- PMID 24218329.
- ^ PMID 30622262.
- ^ PMID 26089148.
- PMID 22366038.
- PMID 16131835.
- PMID 16679312.
External links
- Polyamines in cell cycle proliferation and cell death
- Kilpeläinen, Pekka (2002). Expression and regulation in rat brain and in transgenic mice. Department of Biochemistry, University of Oulu. ISBN 951-42-6631-5. Extensive review of literature through 2001 on polyamine structure, properties, metabolism in mammals, and physiological and pathophysiological roles (See article Table of Contents)