Galantamine total synthesis
The natural source of galantamine are certain species of
Outline
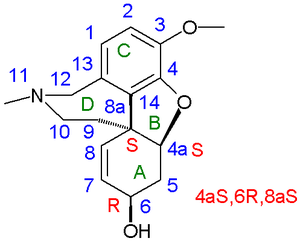
In 1962
In 1989 Vlahov exploited asymmetric reduction by
 |
| Resolution of Narwedine |
|---|
In 1999 Jordis performed (−)-galanthamine synthesis on a multikilogram scale based on Carrol chemistry and Shieh/Carlson chiral resolution. This would become the basis for current industrial production by Sanochemia (AT). In 2000 Fels proposed an
Syntheses of racemic galanthamine have been reported by Wang in 2006 [9] and by Saito in 2008.[10]
Sanochemia industrial production
The method outlined by Jordis in 1999 forms the basis for industrial galanthamine production.[11]

|

| |
| Narwedine synthesis part A | Narwedine synthesis part B |
This method is based on
Enantiopure (−)-narwedine is obtained via the dynamic chiral resolution method pioneered by Shieh/Carlson and in the final step the ketone is reduced to the alcohol with L-selectride.
 |
| reduction of (−)-narwedine to (−)-galanthamine as the bromide |
|---|
This final step is
Trost Galanthamine synthesis
The
 |
| Trost 2005 Galanthamine total synthesis |
|---|
Eli Lilly / U. of Southampton Galanthamine synthesis
A total synthesis reported by

|

| |
| Galanthamine synthesis 2007 A | Galanthamine synthesis 2007 part B |
References and notes
- doi:10.3987/R-1977-09-1752 (inactive 2024-02-17).)
{{cite journal}}: CS1 maint: DOI inactive as of February 2024 (link - doi:10.3987/S(S)-1977-01-0277 (inactive 2024-02-17).)
{{cite journal}}: CS1 maint: DOI inactive as of February 2024 (link - PMID 18629982.
- ^ PMID 17429978.
- PMID 19835379.
- doi:10.3987/COM-10-S(E)27 (inactive 2024-02-17). Archived from the original on 2011-07-22.)
{{cite journal}}: CS1 maint: DOI inactive as of February 2024 (link - PMID 16623560.
- PMID 18781800.
- DIBAL-H, m aqueous NaH2PO4 n NaCNBH3
