Globo H
| |
| Names | |
|---|---|
| IUPAC name
N-((2S,3R,E)-1-(((2R,3R,4R,5S,6R)-5-(((2S,3R,4R,5R,6R)-5-(((2R,3R,4S,5S,6R)-4-(((2S,3R,4R,5R,6R)-3-acetamido-4-(((2R,3R,4S,5R,6R)-4,5-dihydroxy-6-(hydroxymethyl)-3-(((2S,3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)tetrahydro-2H-pyran-2-yl)oxy)-5-hydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3,5-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3,4-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3,4-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3-hydroxyoctadec-4-en-2-yl)palmitamide
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| |
| |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Globo H (globohexaosylceramide) is a globo-series glycosphingolipid antigen that is present on the outer membrane of some cancer cells.[1][2] Globo H is not expressed in normal tissue cells, but is expressed in a number of types of cancers, including cancers of the breast, prostate, and pancreas.[1][3] Globo H's exclusivity for cancer cells makes it a target of interest for cancer therapies.[1][2]
Structure
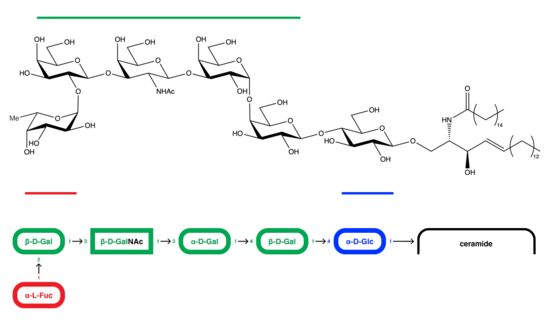
Defined by the monoclonal antibody MBr1, Globo H has been isolated from breast cancer cell line MCF-7, and its structure has been determined through several analyses, including NMR spectroscopy and methylation analysis.[5] Globo H consists of a hexasaccharide of the structure Fucα(1-2)Galβ(1-3)GalNAcβ(1-3)Galα(1-4)Galβ(1-4)Glcβ(1) with a ceramide attached to its terminal glucose ring at the 1 position in a beta linkage.[6]
Synthesis
Biosynthesis

Globo H's biosynthetic pathway is involved in the synthesis pathways of other globo-series glycosphingolipid antigens that are also specific to cancer cells, including stage-specific embryonic antigen-3 (SSEA3) and stage-specific embryonic antigen-4 (SSEA4).[1] The biosynthetic pathway of these antigens includes the enzyme β 1,3-galactosyltransferase V (β3GalT5).[1] β3GalT5 catalyzes the galactosylation of globoside-4 (Gb4) to SSEA3.[1] SSEA3 can then be converted to SSEA4 by sialyltransferase adding a sialic acid group to its end, or it can be converted to Globo H by fucosyltransferase adding a fucose ring to its end.[1] Playing a part in the formation of three different cancer-specific antigens, β3GalT5 is of particular interest in its relevance to cancer treatment, and it has been shown to be critical for cancer cell survival.[8]
Chemical Synthesis
In order to study its potential as a cancer therapy target, Globo H has been synthesized in the laboratory.[9] One synthesis is achieved by first building two trisaccharides from their component sugars, and then linking them.[9] The trisaccharides, with most of their functional groups protected to prevent side reactions, are linked by creating the GalNAcβ(1-3)Gal bond.[9] A thioethyl group is added to the 1 position on one of the protected galactose rings, and in the presence of methyl triflate, this reacts with the hydroxyl group on the 3 position of the other galactose to link the trisaccharides and form the hexasaccharide.[9] The ceramide is added to the 1 position of the terminal glucose ring after hexasaccharide formation.[9]
Globo H as a Therapeutic Target
As a Tumor Associated Carbohydrate Antigen (TACA), Globo-H is a promising clinical target for immunotherapy. While absent in normal tissues, the
Globo H Anticancer Vaccines
Globo-H's TACA character allows for its utilization as an anticancer vaccine, inducing antibody response against the epitope. The resulting humoral immunity could enable the selective eradication of Globo H-presenting tumors.[12] The Taiwanese biopharma company OBI Pharma, Inc., was first to develop Adagloxad Simolenin (OBI-822), a Globo H hexasaccharide conjugated with the immunostimulatory carrier protein KLH.[12] The Phase III GLORIA study is underway evaluating the carbohydrate-based immunogen's effects in high risk triple-negative breast cancer (TNBC) patients with an estimated completion date in 2027.[13]
Alternative vaccine conjugates have been developed which avoid issues associated with the protein carrier KLH by substituting it with a lipid or carbohydrate-based carrier. Examples include the use of lipid A derivatives[14] or entirely carbohydrate vaccine conjugates such as Globo H-PS A1[15]
Anti-Globo H Antibodies
Globo H-targeting antibodies are another strategy currently being evaluated in the cancer therapeutic space. OBI Pharma's OBI-888 is a humanized
Based on OBI-888, the first-in-class
References
- ^ PMID 30808745.
- ^ )
- PMID 29036791.
- ISSN 0002-7863.
- PMID 6501317.
- PMID 18689688.
- PMID 32512916.
- PMID 26677875.
- ^ ISSN 0002-7863.
- S2CID 22303340.
- )
- ^ PMID 25665650.
- S2CID 219778189.
- PMID 26918109.
- PMID 34123241.
- )
- )
- ^ OBI Pharma, Inc (2020-08-04). "A Phase I/II, Open-Label, Dose Escalation and Cohort Expansion Study Evaluating the Safety, Pharmacokinetics (PK), Pharmacodynamics (PD), and Therapeutic Activity of OBI-888 in Patients With Locally Advanced or Metastatic Solid Tumors".
{{cite journal}}: Cite journal requires|journal=(help) - )
- S2CID 219773934.
- ^ Inc, OBI Pharma. "OBI Pharma Granted U.S. FDA Orphan Drug Designation for the Treatment of Gastric Cancer for Its Antibody-Drug Conjugate (ADC) Targeted Cancer Therapy, OBI-999". www.prnewswire.com (Press release). Retrieved 2021-02-27.
{{cite press release}}:|last=has generic name (help)
