Lanicemine
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
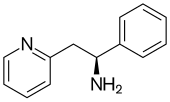
Lanicemine (AZD6765) is a low-trapping NMDA receptor antagonist that was under development by AstraZeneca for the management of severe and treatment-resistant depression.[1][2] Lanicemine differs from ketamine in that it is a low-trapping NMDA receptor antagonist, showing similar rapid-acting antidepressant effects to ketamine in clinical trials but with little or no psychotomimetic side effects.[3] However, lanicemine did not meet study endpoints, and its development was terminated by AstraZeneca in 2013.[4]
See also
- 4-Chlorokynurenine
- AD-1211
- Apimostinel
- CERC-301
- Diphenidine
- Ephenidine
- Esketamine
- Lefetamine
- Memantine
- Methoxphenidine
- MT-45
- Rapastinel
References
- ^ "Lanicemine". AdisInsight. Retrieved 18 June 2017.
- PMID 26724279.
- PMID 23206319.
- ^ Flowers S. "Return to growth: AstraZeneca's CEO Pascal Soriot says 2013 was year of "momentum" for the company". Retrieved 6 February 2014.
