Litmus
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|


Litmus is a
History
The word "litmus" comes from an Old Norse word for “moss used for dyeing”.[1] About 1300, the Spanish physician Arnaldus de Villa Nova began using litmus to study acids and bases.[2][3]
From the 16th century onwards, the blue dye was extracted from some lichens, especially in the Netherlands.
Natural sources

Litmus can be found in different species of
Uses
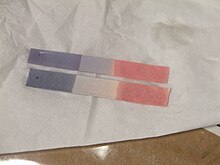
The main use of litmus is to test whether a solution is
Litmus can also be prepared as an aqueous solution that functions similarly. Under acidic conditions, the solution is red, and under alkaline conditions, the solution is blue.
| Litmus (pH indicator) | ||
| below pH 4.5 | above pH 8.3 | |
| 4.5 | ⇌ | 8.3 |
Chemical reactions are other than acid–base can also cause a color change to litmus paper. For instance, chlorine gas turns blue litmus paper white; the litmus dye is bleached[4] because hypochlorite ions are present. This reaction is irreversible, so the litmus is not acting as an indicator in this situation.
Chemistry
The litmus mixture has the
A recipe to make litmus out of the lichens, as outlined on a UC Santa Barbara website says:[8]
Details are difficult to find because the processes were kept secret.
This summary of a modern manufacturing procedure is from The Vanishing Lichens, D H S Richardson, London, 1975. The lichens (preferably
.Stir the lichens from time to time and the color changes from red to purple and finally blue after about four weeks. The lichens are then dried and powdered. At this stage the lichens contain partly litmus and partly orcein pigments. The orcein is removed by extraction with alcohol, leaving the pure blue litmus. It is marketed as blue lumps, masses, or tablets, after mixing with colorless compounds such as chalk and gypsum. Litmus paper is paper impregnated with this substance.
Mechanism
Red litmus contains a weak
References
- ^ "Litmus". 7 October 2021.
- ^ a b c Neupert, Manfred (January 31, 2013). "Lackmus". Römpp Lexikon Chemie (in German).
- ^
Surber, Christian; Humbert, Philippe; Abels, Christoph; ISBN 9783318063851. Retrieved 1 May 2023.
In the early 14th century, the Spanish scholar, Arnaldus de Villa Nova (1235–1311), began to use litmus for studying acids and bases. This compound, extracted from a lichen, had been used as a dye since at least the time of the Vikings, but he was the first person known to use it as a test of acidity.
- ^ O'Leary, Donal (2000). "Chlorine". The Chemical Elements. Archived from the original on 2008-12-21.
- S2CID 41944320.
- .
- ^ E. T. Wolf: Vollständige Übersicht der Elementar-analytischen Untersuchungen organischer Substanzen, S. 450–453, veröffentlicht 1846, Verlag E. Anton (Germany).
- ^ "Students ask questions, UCSB scientists answer them". UCSB ScienceLine. 2002.
