Meso compound
A meso compound or meso isomer is an
For example,
It is a requirement for two of the stereocenters in a meso compound to have at least two substituents in common (although having this characteristic does not necessarily mean that the compound is meso). For example, in 2,4-pentanediol, both the second and fourth carbon atoms, which are stereocenters, have all four substituents in common.

Since a meso isomer has a superposable mirror image, a compound with a total of n chiral centers cannot attain the theoretical maximum of 2n stereoisomers if one of the stereoisomers is meso.[4]
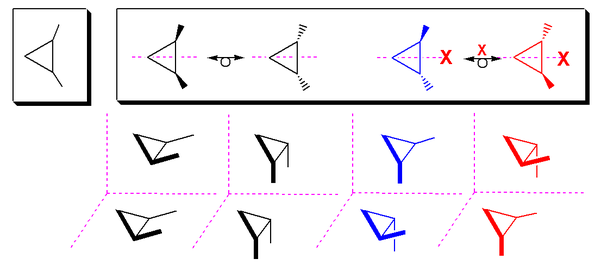
A meso isomer need not have a mirror plane. It may have an
Cyclic meso compounds
1,2-substituted
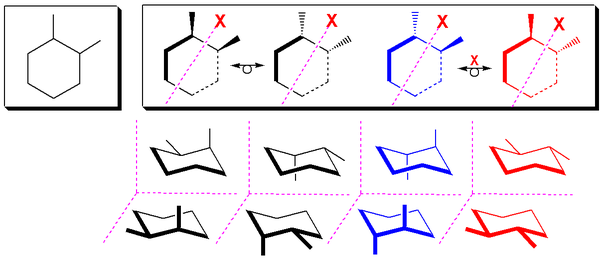
The two cis stereoisomers of 1,2-substituted cyclohexanes behave like meso compounds at room temperature in most cases. At room temperature, most 1,2-disubstituted cyclohexanes undergo rapid ring flipping (exceptions being rings with bulky substituents), and as a result, the two cis stereoisomers behave chemically identically with chiral reagents.[5] At low temperatures, however, this is not the case, as the activation energy for the ring-flip cannot be overcome, and they therefore behave like enantiomers. Also noteworthy is the fact that when a cyclohexane undergoes a ring flip, the absolute configurations of the stereocenters do not change.
References
- ISSN 0021-9584.
- ^ ISBN 978-0-495-11258-7.
- ^ Bruice, Paula. Organic Chemistry. 2007. Pearson Prentice Hall. Upper Saddle River NJ.
- ^ Vollhardt, K. Peter C. Organic Chemistry: Structure and Function, Fourth Ed. 2003. W.H. Freeman and Co. New York. pp. 187.