Organocatalysis
In organic chemistry, organocatalysis is a form of catalysis in which the rate of a chemical reaction is increased by an organic catalyst. This "organocatalyst" consists of carbon, hydrogen, sulfur and other nonmetal elements found in organic compounds.[3][4][5][6][7][8] Because of their similarity in composition and description, they are often mistaken as a misnomer for enzymes due to their comparable effects on reaction rates and forms of catalysis involved.
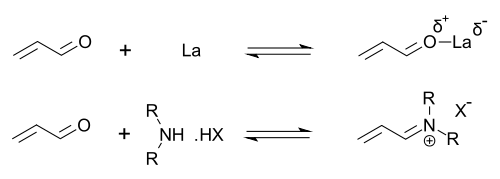
Organocatalysts which display secondary amine functionality can be described as performing either enamine catalysis (by forming catalytic quantities of an active enamine nucleophile) or iminium catalysis (by forming catalytic quantities of an activated iminium electrophile). This mechanism is typical for covalent organocatalysis. Covalent binding of substrate normally requires high catalyst loading (for proline-catalysis typically 20–30 mol%). Noncovalent interactions such as hydrogen-bonding facilitates low catalyst loadings (down to 0.001 mol%).
Organocatalysis offers several advantages. There is no need for metal-based catalysis thus making a contribution to
Introduction
Regular achiral organocatalysts are based on nitrogen such as
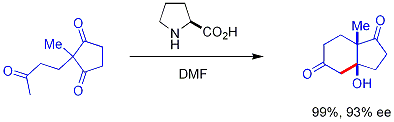
In this reaction, naturally occurring chiral proline is the chiral catalyst in an Aldol reaction. The starting material is an achiral triketone and it requires just 3% of proline to obtain the reaction product, a ketol in 93% enantiomeric excess. This is the first example of an amino acid-catalyzed asymmetric aldol reaction.[16][17]
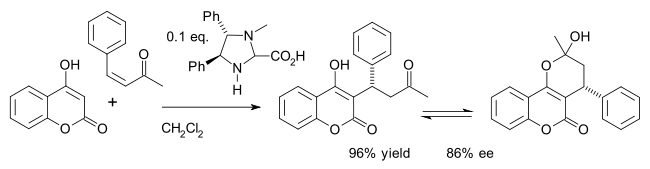
The asymmetric synthesis of the
Many chiral organocatalysts are an adaptation of
A breakthrough in the field of organocatalysis came in 1997 when Yian Shi reported the first general, highly enantioselective organocatalytic reaction with the catalytic asymmetric epoxidation of trans- and trisubstituted olefins with chiral dioxiranes.[20] Since that time, several different types of reactions have been developed.
Organocatalyst classes
Organocatalysts for asymmetric synthesis can be grouped in several classes:
- cinchona alkaloids, certain oligopeptides.
- Synthetic catalysts derived from biomolecules.
- Triazolium salts as next-generation Stetter reaction catalysts
Examples of asymmetric reactions involving organocatalysts are:
- Asymmetric Diels-Alder reactions
- Asymmetric Michael reactions
- Asymmetric Mannich reactions
- Shi epoxidation
- Organocatalytic transfer hydrogenation
Proline
Proline catalysis has been reviewed.[22][23]
Imidazolidinone organocatalysis
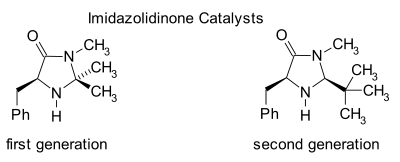
The transient iminium intermediate is chiral which is transferred to the reaction product via
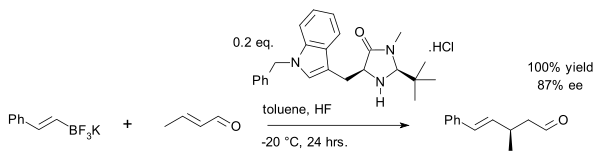
One example is the asymmetric synthesis of the drug
A recent exploit is the vinyl alkylation of crotonaldehyde with an organotrifluoroborate salt:[27]
For other examples of its use: see
Thiourea organocatalysis
A large group of organocatalysts incorporate the
Their current uses are restricted to asymmetric multicomponent reactions, including those involving Michael addition, asymmetric multicomponent reactions for the synthesis of spirocycles, asymmetric multicomponent reactions involving acyl Strecker reactions, asymmetric Petasis reactions, asymmetric Biginelli reactions, asymmetric Mannich reactions, asymmetric aza-Henry reactions, and asymmetric reductive coupling reactions.[29]
References
- .
- .
- ISBN 978-3-527-30517-9.
- .
- PMID 15455437.
- PMID 17198969.
- PMID 17225236.
- PMID 11668532.
- ^ International Patent WO 2006068611 A1 20060629 " Direct Homogeneous and Heterogeneous Organic Acid and Amino Acid-Catalyzed Modification of Amines and Alcohols" Inventors: Armando Córdova, Stockholm, Sweden; Jonas Hafrén, Stockholm, Sweden.
- ^ Example 4 in U.S. Patent 3,975,440 August 17, 1976, Filed Dec. 9, 1970 Zoltan G. Hajos and David R. Parrish.
- ^ "2021 Nobel Prize in chemistry". Nobel Prize. Nobel Prize. Retrieved 6 October 2021.
- PMID 20175175.
- .
- PMID 12630854.
- S2CID 205215034.
- ^ Z. G. Hajos, D. R. Parrish, German Patent DE 2102623 1971
- .
- .
- .
- ISSN 0002-7863.
- PMID 19623342.
- PMID 17198969.
- S2CID 93168492.
- ^ Gérald Lelais; David W. C. MacMillan (2006). "Modern Strategies in Organic Catalysis: The Advent and Development of Iminium Activation" (PDF). Aldrichimica Acta. 39 (3): 79.
- PMID 18072802.
- PMID 14579449.
- S2CID 34848947.
- .
- .
External links
 Media related to Organocatalysis at Wikimedia Commons
Media related to Organocatalysis at Wikimedia Commons The dictionary definition of organocatalysis at Wiktionary
The dictionary definition of organocatalysis at Wiktionary Quotations related to Organocatalysis at Wikiquote
Quotations related to Organocatalysis at Wikiquote