Nef reaction
| Nef reaction | |
|---|---|
| Named after | John Ulric Nef |
| Reaction type | Substitution reaction |
| Identifiers | |
| Organic Chemistry Portal | nef-reaction |
| RSC ontology ID | RXNO:0000157 |
In
nitroalkane (R−NO2) to an aldehyde (R−CH=O) or a ketone (R2C=O) and nitrous oxide (N2O). The reaction has been the subject of several literature reviews.[1][2][3]
The reaction was reported in 1894 by the chemist
yield of nitrous oxide and at least 70% yield of acetaldehyde. However, the reaction was pioneered a year earlier in 1893 by Konovalov,[5] who converted the potassium salt of 1-phenylnitroethane with sulfuric acid to acetophenone
.
Reaction mechanism
The
resonance structures
1a and 1b is depicted below:
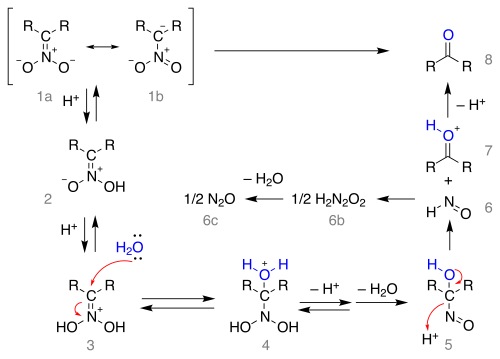
The salt is protonated forming the
carbonyl
compound.
Note that formation of the nitronate salt from the nitro compound requires an
alpha hydrogen
atom and therefore the reaction fails with tertiary nitro compounds.
Scope
Nef-type reactions are frequently encountered in
Michael reaction in the synthesis of the γ-keto-carbonyl methyl 3-acetyl-5-oxohexanoate, itself a cyclopentenone intermediate:[7][8]

In
isotope labeling of C14-D‑mannose and C14-D‑glucose from D‑arabinose and C14‑nitromethane (the first step here is a Henry reaction
):

The opposite reaction is the Wohl degradation.
Variants
Nef's original protocol, using concentrated
Oxophilic reductants, such as titanium salts, will reduce the nitronate to a hydrolysis-susceptible imine, but less selective reductants give the amine instead.[9]
References
- .
- ISBN 9780471515944.
- ISBN 9780080359298.
- .
- ^ Konovalov.,: J. Russ. Phys. Chem. Soc. 2 1893, 6(I), 509.
- ^ Warren & Wyatt 2008, p. 161.
- .
- ^ a b Warren, Stuart; Wyatt, Paul (2008). Organic Synthesis: the disconnection approach (2nd ed.). Wiley. pp. 161–164.
- .
- .
- .
