Nylon 6
| |
| Names | |
|---|---|
| IUPAC name
Poly(azepan-2-one); poly(hexano-6-lactam)
| |
| Systematic IUPAC name
Poly[azanediyl(1-oxohexane-1,6-diyl)] | |
| Other names
Polycaprolactam, polyamide 6, PA6, poly-ε-caproamide, Perlon, Dederon, Capron, Ultramid, Akulon, Nylatron, Kapron, Alphalon, Tarnamid, Akromid, Frianyl, Schulamid, Durethan, Technyl, Nyorbits ,Winmark Polymers
| |
| Identifiers | |
| ChemSpider |
|
ECHA InfoCard
|
100.124.824 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C6H11NO)n | |
| Density | 1.084 g/mL [citation needed] |
| Melting point | 218.3 °C (493 K) |
| Hazards | |
| 434 °C; 813 °F; 707 K | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nylon 6 or polycaprolactam is a
History
Polycaprolactam was developed by
The Soviet Union began its development of an analog in the 1940s, while negotiating with Hitler on building an IG Farben plant in Ukraine, basic scientific work was ongoing in 1942. The production only started in 1948 in Klin, after USSR got its hands on the 2000 volumes of IG Farben, and 10,000 volumes of AEG technical documentation,[2] as a result of victory in the World War II.
Synthesis
Nylon 6 can be modified using comonomers or stabilizers during polymerization to introduce new chain end or functional groups, which changes the reactivity and chemical properties. It is often done to change its dyeability or flame retardance.[3] Nylon 6 is synthesized by ring-opening polymerization of caprolactam. Caprolactam has 6 carbons, hence Nylon 6. When caprolactam is heated at about 533 K in an inert atmosphere of nitrogen for about 4–5 hours, the ring breaks and undergoes polymerization. Then the molten mass is passed through spinnerets to form fibres of nylon 6.

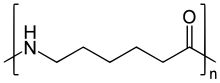
During polymerization, the amide bond within each caprolactam molecule is broken, with the active groups on each side re-forming two new bonds as the monomer becomes part of the polymer backbone. Unlike nylon 6,6, in which the direction of the amide bond reverses at each bond, all nylon 6 amide bonds lie in the same direction (see figure: note the N to C orientation of each amide bond).

Properties
Nylon 6 fibres are tough, possessing high tensile strength, elasticity and lustre. They are wrinkleproof and highly resistant to abrasion and chemicals such as acids and alkalis. The fibres can absorb up to 2.4% of water, although this lowers tensile strength. The glass transition temperature of Nylon 6 is 47 °C.
As a synthetic fibre, Nylon 6 is generally white but can be dyed in a solution bath prior to production for different color results. Its
Biodegradation
Flavobacterium sp. [85] and Pseudomonas sp. (NK87) degrade
Production in Europe
At present, polyamide 6 is a significant construction material used in many industries, for instance in the automotive industry, aircraft industry, electronic and electrotechnical industry, clothing industry and medicine. Annual demand for polyamides in Europe amounts to a million tonnes. They are produced by all leading chemical companies.
The largest producers of polyamide 6 in Europe:[7]
- Fibrant, 260,000 tonnes per year
- BASF, 240,000 tonnes per year
- Lanxess, 170,000 tonnes per year
- Radici, 125,000 tonnes per year
- DOMO, 100,000 tonnes per year
References
- ISBN 978-1469615103
- ISBN 978-5-8243-0882-2.
- ^ "Synthesis of Modified Polyamides (Nylon 6)", NPTEL (National Programme On Technology Enhanced Learning), retrieved May 9, 2016
- ^ ”Polyamide Fiber Physical and Chemical Properties of Nylon 6”, textilefashionstudy.com, retrieved May 9, 2016.
- PMID 19865515.
- ^ https://scitechdaily.com/new-catalyst-completely-breaks-down-durable-plastic-pollution-in-minutes/
- ^ "Segment Tworzywa 2015" (PDF) (in Polish). static.grupaazoty.com. Retrieved 2016-04-12.
- ^ "Alphalon™ (PA6)" (in Polish). att.grupaazoty.com. Archived from the original on 2016-04-26. Retrieved 2016-04-12.
- ^ "Grupa Azoty: Nowa wytwórnia pozwoli zająć pozycję 2. producenta poliamidu w UE" (in Polish). wyborcza.biz. Retrieved 2016-04-12.
