Onapristone
Appearance
 | |
| Clinical data | |
|---|---|
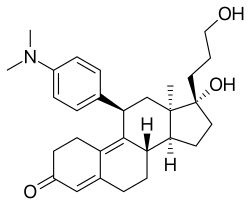
| Other names | ZK-89299; ZK-299; AR-18; IVV-1001; 11β-(4-(Dimethylamino)phenyl)-17α-hydroxy-17β-(3-hydroxypropyl)-13α-estra-4,9-dien-3-one |
| Drug class | Antiprogestogen |
| Identifiers | |
| |
JSmol) | |
| |
| |
Onapristone (
contraceptive, but was discontinued during phase III clinical trials in 1995 due to findings that liver function abnormalities developed in a majority patients.[5][6][7]
Onapristone has been found to be effective in the treatment of breast cancer.[8][5][9]
As of 2016, onapristone has re-emerged and is under development for the treatment of prostate cancer, currently in phase II clinical trials.[10] It was also under development for the treatment of endometrial cancer, breast cancer, ovarian cancer, and uterine cancer, but was discontinued for these indications in favor of focusing on prostate cancer.[10]
See also
- List of investigational sex-hormonal agents § Progestogenics
- Aglepristone
- Lilopristone
- Telapristone
- Toripristone
