Phloretin
| |
| Names | |
|---|---|
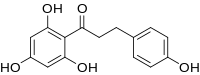
| Preferred IUPAC name
3-(4-Hydroxyphenyl)-1-(2,4,6-trihydroxyphenyl)propan-1-one | |
| Other names
Dihydronaringenin
Phloretol | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.000.444 |
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H14O5 | |
| Molar mass | 274.272 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phloretin is a dihydrochalcone, a type of natural phenol. It can be found in apple tree leaves[1] and the Manchurian apricot.[2]
Metabolism
In rats, ingested phlorizin is converted into phloretin by hydrolytic enzymes in the small intestine.[3][4] Phloretin hydrolase hydrolyses phloretin into phloretic acid and phloroglucinol.
Pharmacological research
In an animal model, phloretin inhibited
aquaporin 9 (AQP9) on mouse hepatocytes.[9]
Nanoparticle Synthesis
Phloretin functionalized gold-nanoparticles (Pht-GNPs) were synthesized using a single-step synthesis method and tested for its anticancer activity. Pht-GNPs showed significant cancer cell toxicities compared to free phloretin.[10]
Glycosides
- Phlorizin is the 2'-glucoside of phloretin
- Naringin dihydrochalcone is a diglycoside of phloretin
See also
- Phloretin-glucosidase
References
- .
- ^ "Manchurian Apricot (Prunus armeniaca var. mandshurica)" (PDF). North Dakota State University. Retrieved January 30, 2014.
- ^ PMID 19125776.
- ^ PMID 11739871.
- PMID 14019989. Retrieved 2012-10-21.
- PMID 15123796.
- S2CID 22143248. Archived from the originalon 2016-03-04. Retrieved 2012-10-21.
- S2CID 3638970.
- PMID 15123796.
- PMID 29636609.
