Radical initiator
In
organic and inorganic peroxides.[2]
Main types of initiation reaction
- Halogens undergo ultraviolet light. This process is used for chlorination of alkanes.
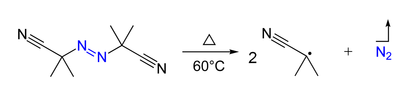
- Azo compounds (R-N=N-R') can be the precursor of two carbon-centered radicals (R• and R'•) and nitrogen gas upon heating and/or by irradiation. For example, AIBN and ABCN yield isobutyronitrile and cyclohexanecarbonitrile radicals, respectively.
- PhC)OO)2) generates benzoyloxyl radicals (PhCOO•), each of which loses carbon dioxide to be converted into a phenyl radical (Ph•). Methyl ethyl ketone peroxide is also common, and acetone peroxideis on rare occasions used as a radical initiator, too.
- Inorganic peroxides function analogously to organic peroxides. Many polymers are often produced from the alkenes upon initiation with peroxydisulfate salts. In solution, peroxydisulfate dissociates to give sulfate radicals:[3]
- [O3SO-OSO3]2− ⇌ 2 [SO4]−
The sulfate radical adds to an alkene forming radical sulfate esters, e.g. .CHPhCH2OSO3−, that add further alkenes via formation of C-C bonds. Many styrene and fluoroalkene polymers are produced in this way.
- In catalyst.

Safety
Some radical initiators such as azo compounds and peroxides can detonate at elevated temperatures so they must be stored cold.
References
- OCLC 642506595
- ISBN 978-0-471-72091-1
- ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link