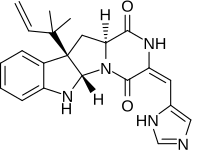
Roquefortine C
| |
| Names | |
|---|---|
| Preferred IUPAC name
(3E,5aS,10bR,11aS)-3-[(1H-Imidazol-5-yl)methylidene]-10b-(2-methylbut-3-en-2-yl)-6,10b,11,11a-tetrahydro-2H-pyrazino[1′,2′:1,5]pyrrolo[2,3-b]indole-1,4(3H,5aH)-dione | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C22H23N5O2 | |
| Molar mass | 389.5 g/mol |
| Appearance | White to off-white solid |
| Soluble in ethanol, methanol, DMF or DMSO | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Roquefortine C is a
Danish Blue, Stilton and Gorgonzola
.
Roquefortine C is a cyclodipeptide mycotoxin derived from the diketopiperazine cyclo(Trp-dehydro-His) and is a relatively common fungal metabolite produced by a number of Penicillium species. It is also considered one of the most important fungal contaminants of carbonated beverages, beer, wine, meats, cheese and bread.
Roquefortine C contains the unusual E-dehydrohistidine moiety, a system that typically undergoes facile
Z-isomer of roquefortine C.[10]

However isoroquefortine C is not a natural product and in contrast to roquefortine C does not bind iron. Both have been synthesised.[10]
Related compounds
References
- PMID 22575049.
- PMID 15734568.
- S2CID 1334464.
- ^ PMID 11559041.
- ^ SCBT. "Roquefortine - A potent neurotoxin produced most notably by Penicillium species".
{{cite journal}}: Cite journal requires|journal=(help) - ^ EPA. "Penicillium roqueforti Final Risk Assessment".
{{cite journal}}: Cite journal requires|journal=(help) - PMID 11271775.
- PMID 2231314.
- S2CID 9880652.
- ^ PMID 18412344.
