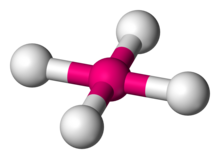
Square planar molecular geometry
| Square planar molecular geometry | |
|---|---|
 | |
| Examples | Xenon tetrafluoride, Potassium tetrachloroplatinate |
| Point group | D4h |
| Coordination number | 4 |
| Bond angle(s) | 90° |
| μ (Polarity) | 0 |

In chemistry, the square planar molecular geometry describes the stereochemistry (spatial arrangement of atoms) that is adopted by certain chemical compounds. As the name suggests, molecules of this geometry have their atoms positioned at the corners.
Examples
Numerous compounds adopt this geometry, examples being especially numerous for transition metal complexes. The noble gas compound xenon tetrafluoride adopts this structure as predicted by VSEPR theory. The geometry is prevalent for transition metal complexes with d8 configuration, which includes Rh(I), Ir(I), Pd(II), Pt(II), and Au(III). Notable examples include the anticancer drugs cisplatin, [PtCl2(NH3)2], and carboplatin. Many homogeneous catalysts are square planar in their resting state, such as Wilkinson's catalyst and Crabtree's catalyst. Other examples include Vaska's complex and Zeise's salt. Certain ligands (such as porphyrins) stabilize this geometry.
Splitting of d-orbitals
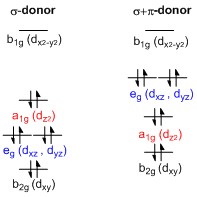
A general
See also
- AXE method
- Molecular geometry
References
- ISSN 0021-9584.
External links
- 3D Chem – Chemistry, Structures, and 3D Molecules
- IUMSC – Indiana University Molecular Structure Center
- Interactive molecular examples for point groups
- [1] – Coordination numbers and complex ions
