Sulfenyl chloride
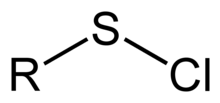
In
Typically, sulfenyl halides are stabilized by
Preparation
Sulfenyl chlorides are typically prepared by
Some
In a variation on the Reed reaction, sulfur dichloride displaces hydrogen under UV light.[8]
Reactions
Perchloromethyl mercaptan (CCl3SCl) reacts with N−H bonds in the presence of base to give the sulfenamides:
This method is used in the production of the fungicides Captan and Folpet.
Sulfenyl chlorides add across alkenes, for example ethylene:[9]
They undergo
Sulfenyl chlorides react with water and
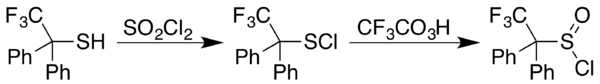
Route to sulfinyl halides
Sulfenyl chlorides can be converted to
Related compounds
Sulfenyl fluorides and bromides are also known.[12] Simple sulfenyl iodides are unknown because they are unstable with respect to the disulfide and iodine:
Sulfenyl iodides can be isolated as stable compounds if they bear alkyl steric protecting groups as part of a cavity-shaped framework, illustrating the technique of kinetic stabilization of a reactive functionality, as in the case of sulfenic acids.[13]
A related class of compounds are the alkylsulfur trichlorides, as exemplified by methylsulfur trichloride, CH3SCl3.[14]
The corresponding selenenyl halides, R−SeCl, are more commonly encountered in the laboratory. Sulfenyl chlorides are used in the production of agents used in the vulcanization of rubber.
References
- ISBN 9781588905307.
- ^ Hubacher, Max H. (1943). "o-Nitrophenylsulfur chloride". Organic Syntheses; Collected Volumes, vol. 2, p. 455.
- ^ a b Douglass, Irwin B.; Norton, Richard V. (1973). "Methanesulfinyl Chloride". Organic Syntheses; Collected Volumes, vol. 5, pp. 709–715.
- .
- .
- .
- .
- ^ Smith (2020), March's Organic Chemistry, rxn. 14-9.
- .
- ISBN 9780080423234.
- ^ Reno, Daniel S.; Pariza, Richard J. (1998). "Phenyl Vinyl Sulfide". Organic Syntheses; Collected Volumes, vol. 9, p. 662.
- .
- ISBN 9781588905307.




![{\displaystyle {\ce {R-SCl + Cl2 -> [R-SCl2]Cl}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/deaf1569a9b4e7730eaea75337aa67878db5868c)


