Ester
In
Esters can be formed from
Organyl esters of carboxylic acids typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in
There are compounds in which an acidic hydrogen of acids mentioned in this article are not replaced by an organyl, but by some other group. According to some authors, those compounds are esters as well, especially when the first carbon atom of the organyl group replacing acidic hydrogen, is replaced by another atom from the
Nomenclature
Etymology
The word ester was coined in 1848 by a German chemist Leopold Gmelin,[6] probably as a contraction of the German Essigäther, "acetic ether".
IUPAC nomenclature
The names of esters that are formed from an alcohol and an acid, are derived from the parent alcohol and the parent acid, where the latter may be organic or inorganic. Esters derived from the simplest

The chemical formulas of organic esters formed from carboxylic acids and alcohols usually take the form RCO2R' or RCOOR', where R and R' are the
Cyclic esters are called
Orthoesters
An uncommon class of esters are the
.Esters of inorganic acids
Esters can also be derived from inorganic acids.
- Perchloric acid forms perchlorate esters, e.g., methyl perchlorate (CH3−O−Cl(=O)3)
- Sulfuric acid forms sulfate esters, e.g., dimethyl sulfate ((CH3−O−)2S(=O)2) and methyl bisulfate (CH3−O−S(=O)2−OH)
- (CH(−O−NO2)(−CH2−O−NO2)2)
- (O=P(−O−CH3)(−OH)2)
- Carbonic acid forms carbonate esters, e.g. dimethyl carbonate ((CH3−O−)2C=O) and 5-membered cyclic ethylene carbonate ((−CH2−O−)2C=O) (if one classifies carbonic acid as an inorganic compound)
- Trithiocarbonic acid forms trithiocarbonate esters, e.g. dimethyl trithiocarbonate ((CH3−S−)2C=S) (if one classifies trithiocarbonic acid as an inorganic compound)
- Chloroformic acid forms chloroformate esters, e.g. methyl chloroformate (Cl−C(=O)−O−CH3) (if one classifies chloroformic acid as an inorganic compound)
- Boric acid forms borate esters , e.g. trimethyl borate (B(−O−CH3)3)
- Chromic acid forms di-tert-butyl chromate (((CH3)3C−O−)2Cr(=O)2)
Inorganic acids that exist as
- Thiosulfuric acid forms two types of thiosulfate esters, e.g. O,O-dimethyl thiosulfate ((CH3−O−)2S(=O)(=S)) and O,S-dimethyl thiosulfate ((CH3−O−)(CH3−S−)S(=O)2)
- IUPAC
- diethyl phosphonate(H−P(=O)(−O−CH2CH3)2)
Some inorganic acids that are unstable or elusive form stable esters.
- Sulfurous acid, which is unstable, forms stable dimethyl sulfite ((CH3−O−)2S=O)
In principle, a part of metal and metalloid
Structure and bonding
Esters derived from
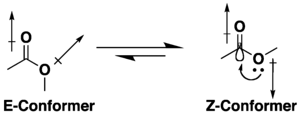
Many carboxylic acid esters have the potential for conformational isomerism, but they tend to adopt an S-cis (or Z) conformation rather than the S-trans (or E) alternative, due to a combination of hyperconjugation and dipole minimization effects. The preference for the Z conformation is influenced by the nature of the substituents and solvent, if present.[9][10] Lactones with small rings are restricted to the s-trans (i.e. E) conformation due to their cyclic structure.
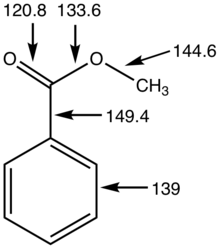
Physical properties and characterization
Esters derived from
Characterization and analysis
Esters are generally identified by gas chromatography, taking advantage of their volatility.
Applications and occurrence
Esters are widespread in nature and are widely used in industry. In nature,
- alpha-linolenic acid (left, red), and oleic acid(top right, blue).
Preparation
Esterification is the general name for a
.Esterification of carboxylic acids with alcohols
The classic synthesis is the
- RCO2H + R'OH ⇌ RCO2R' + H2O
The equilibrium constant for such reactions is about 5 for typical esters, e.g., ethyl acetate.
- Using the alcohol in large excess (i.e., as a solvent).
- Using a dehydrating agent: sulfuric acid not only catalyzes the reaction but sequesters water (a reaction product). Other drying agents such as molecular sievesare also effective.
- Removal of water by physical means such as Dean-Stark apparatus.
Reagents are known that drive the dehydration of mixtures of alcohols and carboxylic acids. One example is the
Another method for the dehydration of mixtures of alcohols and carboxylic acids is the Mitsunobu reaction:
- RCO2H + R'OH + P(C6H5)3 + R2N2 → RCO2R' + OP(C6H5)3 + R2N2H2
Carboxylic acids can be esterified using diazomethane:
- RCO2H + CH2N2 → RCO2CH3 + N2
Using this diazomethane, mixtures of carboxylic acids can be converted to their methyl esters in near quantitative yields, e.g., for analysis by gas chromatography. The method is useful in specialized organic synthetic operations but is considered too hazardous and expensive for large-scale applications.
Esterification of carboxylic acids with epoxides
Carboxylic acids are esterified by treatment with
- RCO2H + RCHCH2O → RCO2CH2CH(OH)R
This reaction is employed in the production of vinyl ester resin from acrylic acid.
Alcoholysis of acyl chlorides and acid anhydrides
Alcohols react with acyl chlorides and acid anhydrides to give esters:
- RCOCl + R'OH → RCO2R' + HCl
- (RCO)2O + R'OH → RCO2R' + RCO2H
The reactions are irreversible simplifying
Alkylation of carboxylic acids and their salts
- RCO2H + (CH3)3OBF4 → RCO2CH3 + (CH3)2O + HBF4
Although rarely employed for esterifications, carboxylate salts (often generated in situ) react with
Transesterification
Transesterification, which involves changing one ester into another one, is widely practiced:
- RCO2R' + CH3OH → RCO2CH3 + R'OH
Like the hydrolysation, transesterification is catalysed by acids and bases. The reaction is widely used for degrading
- n (C6H4)(CO2CH3)2 + 2n C2H4(OH)2 → [(C6H4)(CO2)2(C2H4)]n + 2n CH3OH
A subset of transesterification is the alcoholysis of diketene. This reaction affords 2-ketoesters.[14]
- (CH2CO)2 + ROH → CH3C(O)CH2CO2R
Carbonylation
Alkenes undergo "
- H2C=CH2 + ROH + CO → CH3CH2CO2R
A preparation of methyl propionate is one illustrative example.
- H2C=CH2 + CO + CH3OH → CH3CH2CO2CH3
The carbonylation of methanol yields methyl formate, which is the main commercial source of formic acid. The reaction is catalyzed by sodium methoxide:
- CH3OH + CO → HCO2CH3
Addition of carboxylic acids to alkenes and alkynes
In
- HC≡CH + CH3CO2H → CH3CO2CH=CH2
Vinyl acetate can also be produced by palladium-catalyzed reaction of ethylene, acetic acid, and oxygen:
- 2 H2C=CH2 + 2 CH3CO2H + O2 → 2 CH3CO2CH=CH2 + 2 H2O
Silicotungstic acid is used to manufacture ethyl acetate by the alkylation of acetic acid by ethylene:
- H2C=CH2 + CH3CO2H → CH3CO2CH2CH3
From aldehydes
The
Other methods
- Favorskii rearrangement of α-haloketones in presence of base
- Baeyer–Villiger oxidation of ketones with peroxides
- Pinner reaction of nitriles with an alcohol
- Nucleophilic abstraction of a metal–acyl complex
- Hydrolysis of orthoestersin aqueous acid
- Cellulolysis via esterification[21]
- alcohols.[22]
- ketones leading to methyl esters.[23]
- Interesterification exchanges the fatty acid groups of different esters.
Reactions
Esters react with nucleophiles at the carbonyl carbon. The carbonyl is weakly electrophilic but is attacked by strong nucleophiles (amines, alkoxides, hydride sources, organolithium compounds, etc.). The C–H bonds adjacent to the carbonyl are weakly acidic but undergo deprotonation with strong bases. This process is the one that usually initiates condensation reactions. The carbonyl oxygen in esters is weakly basic, less so than the carbonyl oxygen in amides due to resonance donation of an electron pair from nitrogen in amides, but forms adducts.
Hydrolysis and saponification
Esterification is a reversible reaction. Esters undergo
The alkoxide group may also be displaced by stronger nucleophiles such as ammonia or primary or secondary amines to give amides: (ammonolysis reaction)
- RCO2R' + NH2R″ → RCONHR″ + R'OH
This reaction is not usually reversible. Hydrazines and hydroxylamine can be used in place of amines. Esters can be converted to isocyanates through intermediate hydroxamic acids in the Lossen rearrangement.
Sources of carbon nucleophiles, e.g., Grignard reagents and organolithium compounds, add readily to the carbonyl.
Reduction
Compared to ketones and aldehydes, esters are relatively resistant to reduction. The introduction of catalytic hydrogenation in the early part of the 20th century was a breakthrough; esters of fatty acids are hydrogenated to fatty alcohols.
- RCO2R' + 2 H2 → RCH2OH + R'OH
A typical catalyst is
Especially for fine chemical syntheses,
Direct reduction to give the corresponding ether is difficult as the intermediate hemiacetal tends to decompose to give an alcohol and an aldehyde (which is rapidly reduced to give a second alcohol). The reaction can be achieved using triethylsilane with a variety of Lewis acids.[25][26]
As for
Other reactions
- Methyl esters are often susceptible to decarboxylation in the Krapcho decarboxylation.
- Phenyl esters react to hydroxyarylketones in the Fries rearrangement.
- Specific esters are functionalized with an α-hydroxyl group in the Chan rearrangement.
- Esters with β-hydrogen atoms can be converted to alkenes in ester pyrolysis.
- A direct conversion of esters to nitriles.[27]
- Pairs of esters are coupled to give α-hydroxyketones in the acyloin condensation
Protecting groups
As a class, esters serve as protecting groups for carboxylic acids. Protecting a carboxylic acid is useful in peptide synthesis, to prevent self-reactions of the bifunctional amino acids. Methyl and ethyl esters are commonly available for many amino acids; the t-butyl ester tends to be more expensive. However, t-butyl esters are particularly useful because, under strongly acidic conditions, the t-butyl esters undergo elimination to give the carboxylic acid and isobutylene, simplifying work-up.
Hazards
Esters react with strong
List of ester odorants
Many esters have distinctive fruit-like odors, and many occur naturally in the essential oils of plants. This has also led to their common use in artificial flavorings and fragrances which aim to mimic those odors.
See also
- List of esters
- Amide
- Thioamide
- Carboximidate
- Carbamate
- Xanthate
- Amidine
- Cyanate
- Thiocyanate
- Selenocyanate
- Tellurocyanate
- plastics made of polymericester
- Oligoester, a polymeric ester made of small number of ester monomers
- compressors
- Thioester
- Transesterification
- Ether lipid, an ester that is a lipid and an ether
- Acylal ((R1−C(=O)−O−)(R2−C(=O)−O−)CH−R3)
- orthoboric acid, ...)
- Depside, a polymeric ester, a type of polyphenolic compound composed of two or more monocyclic aromatic units linked by an ester group
- Depsipeptide, a type of ester that is a peptide in which one or more of its amide groups (−C(=O)−NH−) are replaced by the corresponding ester groups (−C(=O)−O−)[29]
- fatty acids and glycerol
- Lactone, a cyclic carboxylic ester
- Lactide, a type of lactone ester
- Vitamin C (ascorbic acid), a lactone ester, an essential nutrient for humans and other animals
- Phthalide, a type of lactone ester
- Coumarin, a type of lactone ester
- Macrolide, a class of natural esters that consist of a large macrocyclic lactone ring to which one or more deoxy sugars may be attached
- Formate
- Chloroformate
References
- ^
- ISBN 9780969054542.
- ISBN 978-1-4200-5045-5.
- ^ "Acetoxytrimethyltin".
- ^ "Trimethyltin acetate | C5H12O2Sn | ChemSpider".
- ^ Leopold Gmelin, Handbuch der Chemie, vol. 4: Handbuch der organischen Chemie (vol. 1) (Heidelberg, Baden (Germany): Karl Winter, 1848), page 182.
Original text:
Translation:b. Ester oder sauerstoffsäure Aetherarten.
Ethers du troisième genre.
Viele mineralische und organische Sauerstoffsäuren treten mit einer Alkohol-Art unter Ausscheidung von Wasser zu neutralen flüchtigen ätherischen Verbindungen zusammen, welche man als gepaarte Verbindungen von Alkohol und Säuren-Wasser oder, nach der Radicaltheorie, als Salze betrachten kann, in welchen eine Säure mit einem Aether verbunden ist.b. Ester or oxy-acid ethers.
Ethers of the third type.
Many mineral and organic acids containing oxygen combine with an alcohol upon elimination of water to [form] neutral, volatile ether compounds, which one can view as coupled compounds of alcohol and acid-water, or, according to the theory of radicals, as salts in which an acid is bonded with an ether. - ^ ISBN 0-471-60180-2.
- ^ "Chemistry of Enols and Enolates – Acidity of alpha-hydrogens". 13 February 2011.
- .
- PMID 12848578.
- .
- ^ Isolation of triglyceride from nutmeg: G. D. Beal "Trimyristen" Organic Syntheses, Coll. Vol. 1, p.538 (1941). Link
- ^ McGee, Harold. On Food and Cooking. 2003, Scribner, New York.
- ^ ISBN 978-3527306732.
- .
- ^ B. Neises & W. Steglich. "Esterification of Carboxylic Acids with Dicyclohexylcarbodiimide/4-Dimethylaminopyridine: tert-Butyl ethyl fumarate". Organic Syntheses; Collected Volumes, vol. 7, p. 93.
- .
- PMID 24770480.
- ISBN 978-3527306732.
- ; Collected Volumes, vol. 1, p. 104.
- S2CID 101737591.
- .
- .
- ^ W. Reusch. "Carboxyl Derivative Reactivity". Virtual Textbook of Organic Chemistry. Archived from the original on 2016-05-16.
- .
- PMID 17602594.
- .
- ^ "PubChem". pubchem.ncbi.nlm.nih.gov. Retrieved 20 April 2023.[failed verification]