tert-Butanesulfinamide
Appearance
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methylpropane-2-sulfinamide | |||
| Identifiers | |||
| |||
3D model (
JSmol ) |
| ||
ECHA InfoCard
|
100.108.188 | ||
PubChem CID
|
|||
| UNII |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| (CH3)3CS(O)NH2 | |||
| Molar mass | 121.20 g/mol | ||
| Appearance | white to off-white crystalline solid | ||
| Melting point | 102 to 105 °C (216 to 221 °F; 375 to 378 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
tert-Butanesulfinamide (also known as 2-methyl-2-propanesulfinamide or Ellman's sulfinamide) is an
asymmetric synthesis as chiral auxiliaries, often as chiral ammonia equivalents for the synthesis of amines.[1][2][3] tert-Butanesulfinamide and the associated synthetic methodology was introduced in 1997 by Jonathan A. Ellman et al.[4]
Enantiopure synthesis
Enantiopure tert-butanesulfinamide can be prepared by enantioselective oxidation of inexpensive di-tert-butyl disulfide to the
3,5-di-tert-butyl salicylaldehyde
.
 |
| tert-Butanesulfinamide synthesis |
|---|
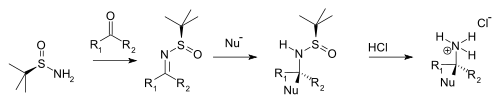
Enantioselective amine synthesis
Condensation with
ammonium salt
or amine (from aldehyde precursor) or the chiral secondary amine (ketone precursor).
 |
| tert-Butanesulfinamide chiral amine synthesis |
|---|
Typical nucleophiles are
organolithium compounds, and enolates
.
Chiral
sulfinimines as intermediates for the asymmetric synthesis of amines have also been developed by Franklin A. Davis.[5]
Applications
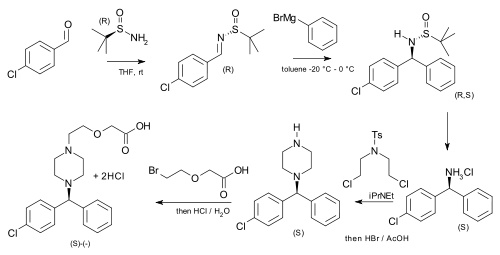
tert-Butanesulfinamide has been used as an auxiliary in an asymmetric synthesis of cetirizine (more potent than the racemic mixture of the drug) starting from p-chlorobenzaldehyde and phenylmagnesium bromide.[6]
 |
| Asymmetric cetirizine synthesis |
|---|


