Thiazyl fluoride

| |

| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NSF | |
| Molar mass | 65.07 g mol−1 |
| Appearance | colourless gas |
| Melting point | −89 °C (−128 °F; 184 K) |
| Boiling point | 0.4 °C (32.7 °F; 273.5 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thiazyl fluoride, NSF, is a colourless, pungent gas at room temperature and condenses to a pale yellow liquid at 0.4 °C.[1] Along with thiazyl trifluoride, NSF3, it is an important precursor to sulfur-nitrogen-fluorine compounds. It is notable for its extreme hygroscopicity.
Synthesis
Thiazyl fluoride can be synthesized by various methods, such as
For synthesis on a preparative scale, the decomposition of compounds already containing the moiety is commonly used:[citation needed]
Reactivity
Reactions with electrophiles and Lewis acids
Lewis acids remove fluoride to afford thiazyl salts:[5]
- NSF + BF3 → [NS]BF4
Thiazyl fluoride functions as a ligand in [Re(CO)5NSF]+.[1] and [M(NSF)6]2+ (M = Co, Ni). In all of its complexes, NSF is bound to the metal center through nitrogen.[6]
Reactions with nucleophiles
Thiazyl fluoride reacts violently with water:[7]
- NSF + 2 H2O → SO2 + HF + NH3

Nucleophilic attack on thiazyl fluoride occurs at sulfur atom:.[8]
- NS−F + Nu− → NS−Nu + F−
Fluoride gives an adduct:
- NS−F + F− → NSF−2
The halogen derivatives XNSF2 (X = F, Cl, Br, I) can be synthesized from reacting Hg(NSF)2 with X2; whereby, ClNSF2 is the most stable compound observed in this series.[9]
Oligomerization and cycloaddition
At room temperature, thiazyl fluoride undergoes cyclic
1,3,5-trifluoro-1,3,5,2,4,6-trithiatriazine is the yielded cyclic trimer, where each sulfur atom remains
Thiazyl fluoride also reacts via exothermic cycloaddition in the presence of dienes.
Structure and bonding

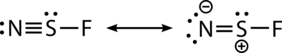
The N−S bond length is 1.448 Å, which is short, indicating multiple bonding, and can be represented by the following resonance structures:
The NSF molecule has 18 total valence electrons and is isoelectronic to sulfur dioxide. Thiazyl fluoride adopts Cs-symmetry and has been shown by isotopic substitution to be bent in the ground state.[10][11] A combination of rotational analysis with Franck-Condon calculations has been applied to study the electronic excitation from the A''A' states, which results in the elongation of the bond by 0.11 Å and a decrease in the NSF by 15.3.