Tetrasulfur tetranitride
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Tetrasulfur tetranitride
| |||
| Systematic IUPAC name
1,3,5,7-tetrathia-2,4,6,8-tetraazacyclooctan-2,4,6,8-tetrayl | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| S4N4 | |||
| Molar mass | 184.287 g/mol | ||
| Appearance | Vivid orange, opaque crystals | ||
| Melting point | 187 °C (369 °F; 460 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Tetrasulfur tetranitride is an inorganic compound with the formula S4N4. This gold-poppy[clarification needed] coloured solid is the most important binary sulfur nitride, which are compounds that contain only the elements sulfur and nitrogen. It is a precursor to many S-N compounds and has attracted wide interest for its unusual structure and bonding.[1][2]
Nitrogen and sulfur have similar
Structure
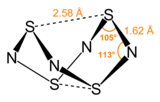
S4N4 adopts an unusual “extreme cradle” structure, with D2d
Properties
S4N4 is stable to
- 2 S4N4 → 4 N2 + S8
Because one of its decomposition products is a gas, S4N4 can be used as an explosive.
Synthesis
S4N4 was first prepared in 1835 by M. Gregory by the reaction of disulfur dichloride with ammonia,[6] a process that has been optimized:[7]
- 6 S2Cl2 + 16 NH3 → S4N4 + S8 + 12 [NH4]Cl
Coproducts of this reaction include heptasulfur imide (S7NH) and elemental sulfur. A related synthesis employs [NH4]Cl instead:[1]
- 4 [NH4]Cl + 6 S2Cl2 → S4N4 + 16 HCl + S8
An alternative synthesis entails the use of (((CH3)3Si)2N)2S as a precursor with pre-formed S–N bonds. (((CH3)3Si)2N)2S is prepared by the reaction of lithium bis(trimethylsilyl)amide and SCl2.
- 2 ((CH3)3Si)2NLi + SCl2 → (((CH3)3Si)2N)2S + 2 LiCl
The (((CH3)3Si)2N)2S reacts with the combination of SCl2 and SO2Cl2 to form S4N4, trimethylsilyl chloride, and sulfur dioxide:[8]
- 2 (((CH3)3Si)2N)2S + 2 SCl2 + 2 SO2Cl2 → S4N4 + 8 (CH3)3SiCl + 2 SO2
Acid-base reactions

S4N4 serves as a
- S4N4 + SbCl5 → S4N4·SbCl5
- S4N4 + SO3 → S4N4·SO3
The reaction of [Pt2Cl4(P(CH3)2
It is protonated by
- S4N4 + H[BF4] → [S4N4H]+[BF4]−
The soft Lewis acid CuCl forms a coordination polymer:[1]
- n S4N4 + n CuCl → (S4N4)n-μ-(−Cu−Cl−)n
Dilute NaOH hydrolyzes S4N4 as follows, yielding thiosulfate and trithionate:[1]
- 2 S4N4 + 6 OH− + 9 H2O → S2O2−3 + 2 S3O2−6 + 8 NH3
More concentrated base yields sulfite:
- S4N4 + 6 OH− + 3 H2O → S2O2−3 + 2 SO2−3 + 4 NH3
Metal complexes
S4N4 reacts with metal complexes. The cage remains intact in some cases but in other cases, it is degraded.
S4N4 as a precursor to other S-N compounds
Many S-N compounds are prepared from S4N4.[10] Reaction with piperidine generates [S4N5]−:
- 24 S4N4 + 32 C5H10NH → 8 [C5H10NH2]+[S4N5]− + 8 (C5H10N)2S + 3 S8 + 8 N2
A related
Treatment with tetramethylammonium azide produces the heterocycle [S3N3]−:
- 8 S4N4 + 8 [(CH3)4N]+[N3]− → 8 [(CH3)4N]+[S3N3]− + S8 + 16 N2
Cyclo-[S3N3]− has 10 pi-electrons.
In a related reaction, the use of the
- 4 S4N4 + 2 [PPN]+[N3]− → 2 [PPN]+[NS4]− + S8 + 10 N2
The anion [NS4]− has a chain structure described using the resonance [S=S=N−S−S−] ↔ [−S−S−N=S=S].
S4N4 reacts with electron-poor alkynes.[11]
Chlorination of S4N4 gives
Passing gaseous S4N4 over
- S4N4 + 8 Ag → 4 Ag2S + 2 N2
- x S4N4 → (SN)4x
Related compounds
- The selenium analogue Se4N4, tetraselenium tetranitride.
Safety
S4N4 is shock-sensitive. Purer samples are more shock-sensitive than those contaminated with elemental sulfur.[7]
References
- ^ a b c d e f g h i Greenwood, N. N.; Earnshaw, A. (1997). Chemical Elements (2nd ed.). Boston, MA: Butterworth-Heinemann. pp. 721–725.
- ^ ISBN 981-256-095-5.
- .
- .
- doi:10.1039/a907106g.
- .
- ^ ISBN 978-0-470-13168-8.
- ISBN 9780471208259.
- .
- ^ ISBN 9780470132562.
- .
- .


