Thioacetal
Appearance
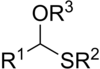

In
analogues of acetals (R−CH(−OR)2). There are two classes: the less-common monothioacetals, with the formula R−CH(−OR')−SR", and the dithioacetals, with the formula R−CH(−SR')2 (symmetric dithioacetals) or R−CH(−SR')−SR" (asymmetric dithioacetals).[1]
The symmetric dithioacetals are relatively common. They are prepared by condensation of thiols (−SH) or dithiols (two −SH groups) with aldehydes (−CH=O). These reactions proceed via the intermediacy of hemithioacetals (R−CH(−OH)−SR'):
- Thiol addition to give hemithioacetal:
- Thiol addition with loss of water to give dithioacetal:
Such reactions typically employ either a
catalyst
.
Dithioacetals generated from aldehydes and either
1,3-propanedithiol are especially common among this class of molecules for use in organic synthesis.[2]
The carbonyl carbon of an aldehyde is
protective groups
for aldehydes.
Far from being unreactive, and in a reaction unlike that of aldehydes, that carbon can be
deprotonated
to render it nucleophilic:
The inversion of polarity between R'(H)Cδ+=Oδ− and R'CLi(SR)2 is referred to as
hydrolyzed back to its carbonyl form. This overall process, the Corey–Seebach reaction
, gives the synthetic equivalent of an acyl anion.




