Uranyl

2

The uranyl ion is an
Structure and bonding

The uranyl ion is linear and symmetrical, with both U–O bond lengths of about 180 pm. The bond lengths are indicative of the presence of multiple bonding between the uranium and oxygen atoms. Since uranium(VI) has the
The uranyl ion is always associated with other ligands. The most common arrangement is for the so-called equatorial ligands to lie in a plane perpendicular to the O–U–O line and passing through the uranium atom. With four ligands, as in [UO2Cl4]2−, the uranium has a distorted
In uranyl fluoride, UO2F2, the uranium atom achieves a coordination number of 8 by forming a layer structure with two oxygen atoms in a uranyl configuration and six fluoride ions bridging between uranyl groups. A similar structure is found in α-uranium trioxide, with oxygen in place of fluoride, except that in that case the layers are connected by sharing oxygen atom from "uranyl groups", which are identified by having relatively short U–O distances. A similar structure occurs in some uranates, such as calcium uranate, CaUO4, which may be written as Ca(UO2)O2 even though the structure does not contain isolated uranyl groups.[3]
Spectroscopy
The colour of uranyl compounds is due to ligand-to-metal charge transfer transitions at ca. 420 nm, on the blue edge of the
Uranyl compounds also exhibit
The uranyl ion has characteristic νU–O stretching
Aqueous chemistry

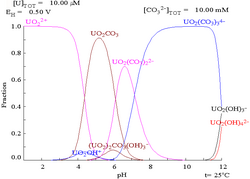
The aqueous uranyl ion is a
As pH increases polymeric species with stoichiometry [(UO2)2(OH)2]2+ and [(UO2)3(OH)5]+ are formed before the hydroxide UO2(OH)2 precipitates. The hydroxide dissolves in strongly alkaline solution to give hydroxo complexes of the uranyl ion.[citation needed]
The uranyl ion can be
Complexes
The uranyl ion behaves as a
2, can be extracted from mixtures containing other ions. Replacing the water molecules that are bound to the uranyl ion in aqueous solution by a second, hydrophobic, ligand increases the solubility of the neutral complex in the organic solvent. This has been called a synergic effect.[14]
The complexes formed by the uranyl ion in aqueous solution are of major importance both in the extraction of uranium from its ores and in nuclear fuel reprocessing. In industrial processes, uranyl nitrate is extracted with tributyl phosphate (TBP, (CH3CH2CH2CH2O)3PO) as the preferred second ligand and kerosene the preferred organic solvent. Later in the process, uranium is stripped from the organic solvent by treating it with strong nitric acid, which forms complexes such as [UO2(NO3)4]2− which are more soluble in the aqueous phase. Uranyl nitrate is recovered by evaporating the solution.[12]
Minerals
The uranyl ion occurs in minerals derived from uranium ore deposits by water-rock interactions that occur in uranium-rich mineral seams. Examples of uranyl containing minerals include:[citation needed]
- silicates: uranophane (H3O)2Ca(UO2)2(SiO4)·3H2O)
- phosphates: autunite (Ca(UO2)2(PO4)2·8–12H2O), torbernite (Cu(UO2)2(PO4)·8–12H2O)
- arsenates: arsenuranospathite (Al(UO2)2(AsO4)2F·20H2O)
- vanadates: carnotite (K2(UO2)2(VO4)2·3H2O), tyuyamunite (Ca(UO2)2V2O8·8H2O)
- carbonates: schröckingerite NaCa3(UO2)(CO3)3(SO4)F·10H2O
- oxalates: uroxite [(UO2)2(C2O4)(OH)2(H2O)2]·H2O.
These minerals are of little commercial value as most uranium is extracted from
Uses
Uranyl salts are used to stain samples for electron and electromagnetic microscopy studies of DNA.[15]
Health and environmental issues
Uranyl salts are toxic and can cause severe
All uranium compounds are
References
- ^ Cotton, S (1991). Lanthanides and Actinides. New York: Oxford University Press. p. 128.
- .
- ISBN 0-19-855125-8.
- S2CID 96229881.
- PMID 16948430.
- PMID 20886130.
- S2CID 94834106.
- PMID 17461564.
- ISBN 0-12-077250-7.
- ISBN 0-471-16392-9.
- ^ "IUPAC SC-Database: A comprehensive database of published data on equilibrium constants of metal complexes and ligands". Academic Software. Archived from the original on 2020-05-09. Retrieved 2011-01-27.
- ^ ISBN 978-0-08-037941-8.
- ^ Neues allgemeines Journal der Chemie (in German). Frölich. 1805.
- .
- PMID 13788706.
- S2CID 25310165.
- PMID 12678382. Archived from the original(PDF) on 2014-01-08. Retrieved 2014-01-08.
- PMID 15511555.
