Uranium trioxide

| |
| Names | |
|---|---|
| IUPAC names
Uranium trioxide
Uranium(VI) oxide | |
| Other names
Uranyl oxide
Uranic oxide | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.014.274 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| UO3 | |
| Molar mass | 286.29 g/mol |
| Appearance | yellow-orange powder |
| Density | 5.5–8.7 g/cm3 |
| Melting point | ~200–650 °C (decomposes) |
| insoluble | |
| Structure | |
| see text | |
| I41/amd (γ-UO3) | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
99 J·mol−1·K−1[1] |
Std enthalpy of (ΔfH⦵298)formation |
−1230 kJ·mol−1[1] |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H300, H330, H373, H411 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
| Uranium dioxide Triuranium octoxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Uranium trioxide (UO3), also called
Production and use
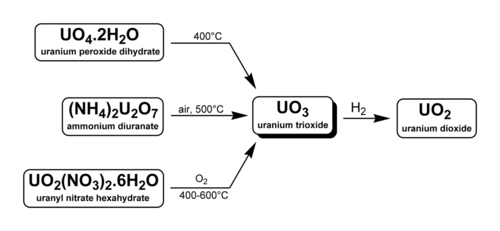
There are three methods to generate uranium trioxide. As noted below, two are used industrially in the reprocessing of nuclear fuel and uranium enrichment.
- U3O8 can be oxidized at 500 °C with oxygen.[2] Note that above 750 °C even in 5 atm O2 UO3 decomposes into U3O8.[3]
- Uranyl nitrate, UO2(NO3)2·6H2O can be heated to yield UO3. This occurs during the reprocessing of nuclear fuel. Fuel rods are dissolved in HNO3 to separate uranyl nitrate from plutonium and the fission products (the PUREX method). The pure uranyl nitrate is converted to solid UO3 by heating at 400 °C. After reduction with hydrogen (with other inert gas present) to uranium dioxide, the uranium can be used in new MOX fuel rods.
- Ammonium diuranate or sodium diuranate (Na2U2O7·6H2O) may be decomposed. Sodium diuranate, also known as yellowcake, is converted to uranium trioxide in the enrichment of uranium. Uranium dioxide and uranium tetrafluoride are intermediates in the process which ends in uranium hexafluoride.[4]
Uranium trioxide is shipped between processing facilities in the form of a gel, most often from mines to conversion plants. When used for conversion, all uranium oxides are often called reprocessed uranium (RepU).[5]
It has been reported that the corrosion of uranium in a silica rich aqueous solution forms
Health and safety hazards
Like all hexavalent uranium compounds, UO3 is hazardous by inhalation, ingestion, and through skin contact. It is a poisonous, slightly radioactive substance, which may cause shortness of breath, coughing, acute arterial lesions, and changes in the chromosomes of
Structure
Solid state structure
The only well characterized binary trioxide of any
Alpha
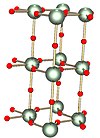 |
The α (alpha) form: a layered solid where the 2D layers are linked by oxygen atoms (shown in red) | Hydrated uranyl peroxide formed by the addition of amorphous form to the alpha form occurs.[15]
|
Beta
 |
The β (beta) UO3 form: This solid contains multiple unique uranium sites and distorted polyhedra. | This form can be formed by heating ammonium diuranate, while P.C. Debets and B.O. Loopstra, found four solid phases in the UO3-H2O-NH3 system that they could all be considered as being UO2(OH)2·H2O where some of the water has been replaced with ammonia.[16][17] It was found that calcination at 500 °C in air forms the beta form of uranium trioxide.[3] Later experiments found the most reliable method for synthesizing pure β-UO3 was to calcinate uranyl nitrate hexahydrate at 450 °C for 6 days and cool slowly over 24 hours.[18] |
Gamma
 |
The γ (gamma) form: with the different uranium environments in green and yellow | The most frequently encountered polymorph is γ-UO3, whose x-ray structure has been solved from powder diffraction data. The compound crystallizes in the space group I41/amd with two uranium atoms in the asymmetric unit. Both are surrounded by somewhat distorted octahedra of oxygen atoms. One uranium atom has two closer and four more distant oxygen atoms whereas the other has four close and two more distant oxygen atoms as neighbors. Thus it is not incorrect to describe the structure as [UO2]2+[UO4]2− , that is uranyl uranate.[19] |
Delta
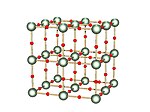 |
The delta (δ) form is a cubic solid where the oxygen atoms are arranged between the uranium atoms.[20]
|
Epsilon
 |
The proposed crystal structure of the epsilon (ε) form consists of sheets of uranium hexagonal bipyramids connected through edge-sharing polyhedra. These sheets are connected through the axial uranyl oxygen atoms. The proposed structure is in the triclinic P-1 space group.[21] |
High pressure form
There is a high-pressure solid form with U2O2 and U3O3 rings in it.[22] [23]
Hydrates
-
Hydrous and anhydrous forms of UO3
-
Anhydrous forms of UO3
Several
Molecular forms
While uranium trioxide is encountered as a polymeric solid under ambient conditions, some work has been done on the molecular form in the gas phase, in matrix isolations studies, and computationally.
Gas phase
At elevated temperatures gaseous UO3 is in
- 2 U3O8(s) + O2(g) ⇌ 6 UO3(g)
With increasing temperature the equilibrium is shifted to the right. This system has been studied at temperatures between 900 °C and 2500 °C. The vapor pressure of monomeric UO3 in equilibrium with air and solid U3O8 at ambient pressure, about 10−5 mbar (1 mPa) at 980 °C, rising to 0.1 mbar (10 Pa) at 1400 °C, 0.34 mbar (34 Pa) at 2100 °C, 1.9 mbar (193 Pa) at 2300 °C, and 8.1 mbar (809 Pa) at 2500 °C.[24][25]
Matrix isolation
Infrared spectroscopy of molecular UO3 isolated in an argon matrix indicates a T-shaped structure (
Computational study

Calculations predict that the point group of molecular UO3 is C2v, with an axial bond length of 1.75 Å, an equatorial bond length of 1.83 Å and an angle of 161° between the axial oxygens. The more symmetrical D3h species is a saddle point, 49 kJ/mol above the C2v minimum. The authors invoke a second-order Jahn–Teller effect as explanation.[27]
Cubic form of uranium trioxide
The crystal structure of a uranium trioxide phase of composition UO2·82 has been determined by X-ray powder diffraction techniques using a Guinier-type focusing camera. The unit cell is cubic with a = 4·138 ± 0·005 kX. A uranium atom is located at (000) and oxygens at (View the MathML source), (View the MathML source), and (View the MathML source) with some anion vacancies. The compound is isostructural with ReO3. The U-O bond distance of 2·073 Å agrees with that predicted by Zachariasen for a bond strength S = 1.[28]
Reactivity
Uranium trioxide reacts at 400 °C with
- 2 CF2Cl2 + UO3 → UF4 + CO2 + COCl2 + Cl2
- 4 CFCl3 + UO3 → UF4 + 3 COCl2 + CCl4 + Cl2
Uranium trioxide can be dissolved in a mixture of tributyl phosphate and thenoyltrifluoroacetone in supercritical carbon dioxide, ultrasound was employed during the dissolution.[30]
Electrochemical modification
The reversible insertion of magnesium cations into the lattice of uranium trioxide by cyclic voltammetry using a graphite electrode modified with microscopic particles of the uranium oxide has been investigated. This experiment has also been done for U3O8. This is an example of electrochemistry of a solid modified electrode, the experiment which used for uranium trioxide is related to a carbon paste electrode experiment. It is also possible to reduce uranium trioxide with sodium metal to form sodium uranium oxides.[31]
It has been the case that it is possible to insert
Uranium oxide is
, depending on the conditions.As an acid
- UO3 + H2O → UO2−
4 + 2 H+
Dissolving uranium oxide in a strong
2O2−
7, or other poly-uranates. Important diuranates include ammonium diuranate ((NH4)2U2O7), sodium diuranate (Na2U2O7) and magnesium diuranate (MgU2O7), which forms part of some yellowcakes. It is worth noting that uranates of the form M2UO4 do not contain UO2−
4 ions, but rather flattened UO6 octahedra, containing a uranyl group and bridging oxygens.[35]
As a base
- UO3 + H2O → UO2+
2 + 2 OH−
Dissolving uranium oxide in a strong acid like
From
Uranium oxides in ceramics
UO3-based ceramics become green or black when fired in a reducing atmosphere and yellow to orange when fired with oxygen. Orange-coloured
Prior to 1960, UO3 was used as an agent of crystallization in crystalline coloured glazes. It is possible to determine with a Geiger counter if a glaze or glass was made from UO3.
References
- ^ ISBN 978-0-618-94690-7.
- .
- ^ .
- .
- ^ "Transport of Radioactive Materials – World Nuclear Association". www.world-nuclear.org. Archived from the original on 5 February 2015. Retrieved 12 April 2018.
- ^ Trueman ER, Black S, Read D, Hodson ME (2003) "Alteration of Depleted Uranium Metal" Goldschmidt Conference Abstracts, p. A493 abstract
- PMID 25964321.
- ^ Schoepite. Webmineral.com. Retrieved on 2011-07-19.
- PMID 22763414.
- PMID 25422465.
- ^ Ander L, Smith B (2002) "Annexe F: Groundwater transport modelling" The health hazards of depleted uranium munitions, part II (London: The Royal Society)
- ^ Smith B (2002) "Annexe G: Corrosion of DU and DU alloys: a brief discussion and review" The health hazards of depleted uranium munitions, part II (London: The Royal Society)
- PMID 15540945.
- .
- .
- .
- S2CID 220746556.
- .
- .
- S2CID 244423124.
- .
- ^ Gmelin Handbuch (1982) U-C1, 129–135.
- .
- .
- .
- .
- .
- .
- .
- .
- .
- .
- .
- ISBN 978-0-19-507366-9.