Whiting event
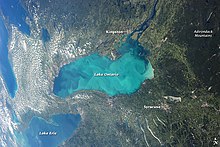
A whiting event is a phenomenon that occurs when a suspended cloud of fine-grained calcium carbonate precipitates in water bodies, typically during summer months, as a result of photosynthetic microbiological activity or sediment disturbance.[1][2][3] The phenomenon gets its name from the white, chalky color it imbues to the water. These events have been shown to occur in temperate waters as well as tropical ones, and they can span for hundreds of meters.[3] They can also occur in both marine and freshwater environments.[4] The origin of whiting events is debated among the scientific community, and it is unclear if there is a single, specific cause. Generally, they are thought to result from either bottom sediment re-suspension or by increased activity of certain microscopic life such as phytoplankton.[5][6][1] Because whiting events affect aquatic chemistry, physical properties, and carbon cycling, studying the mechanisms behind them holds scientific relevance in various ways.[7][2][8][9][10]
Characteristics
Whiting event clouds consist of calcium carbonate
Potential causes
Some debate exists surrounding the exact cause of whiting events. And although much research exists on the subject, there is still no definitive consensus on the chemical mechanisms behind it. The three most common suggested causes for the phenomenon are: microbiological processes, re-suspension of marine or bottom sediments, and spontaneous direct precipitation from water.
Microbiological activity
Substantial findings indicate
Sediment re-suspension
In shallower waters, evidence supports that activity of local fisherman and marine life such as fish and certain shark species can disturb bottom sediments containing calcium carbonate particles and lead to their suspension.[2] In addition, as microorganisms impact water chemistry in observable ways and require certain nutrient levels to thrive, whiting events found occurring in nutrient-poor waters where no significant alkalinity difference exists between whiting and non-whiting waters support the idea of sediment re-suspension as a primary cause.[13]
Relevance
Whiting events have a unique effect on the waters around them. The fact that calcium carbonate clouds increase turbidity and light reflectance holds implications for organisms and processes that depend on light.
References
- ^ a b "Whiting Event, Lake Ontario". NASA Earth Observatory. 2 September 2013.
- ^ S2CID 128695792.
- ^ S2CID 129052529.
- ^ ISSN 0272-7714.
- S2CID 139114.
- ^ "Whiting in Lake Michigan". NASA Earth Observatory. 18 September 2001.
- ^ S2CID 45359827.
- ^ .
- ^ a b Yates, K.K; Robbins, L.L. (2001). "Microbial Lime-Mud Production and Its Relation to Climate Change". AAPG Studies in Geology. Tulsa, Ok: American Association of Petroleum Geologists. pp. 267–283.
- ^ ISSN 1093-474X.
- PMID 36153366.
- ^ S2CID 134734239.
- .
- ISSN 0024-3590.
Further reading
- Thompson, Joel B.; et al. (1997). "Whiting event: Biogenic origin due to the photosynthetic activity of cyanobacterial picoplankton" (PDF). American Society of Limnology and Oceanography. pp. 133–141.
- Effler, Steven W.; Perkins, Mary Gail; Greer, Harry; Johnson, David L. (1987). "Effect of 'whiting' on optical properties and turbidity in Owasco Lake, New York". Journal of the American Water Resources Association. 23 (2): 189–96. .
- "Whiting events (calcium carbonate precipitate), a naturally occurring phenomena" (PDF).

