Willgerodt rearrangement
| Willgerodt rearrangement | |
|---|---|
| Named after | Conrad Willgerodt |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000185 |
The Willgerodt rearrangement or Willgerodt reaction is an
aliphatic
chain (n typically 0 to 5), multiple reactions take place with the amide group always ending up at the terminal end. The net effect is thus migration of the carbonyl group to the end of the chain and oxidation.
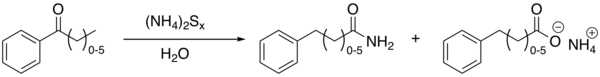
An example with modified reagents (sulfur, concentrated
ammonium hydroxide and pyridine) is the conversion of acetophenone to 2-phenylacetamide and phenylacetic acid[5]

Willgerodt–Kindler reaction
| Willgerodt–Kindler reaction | |
|---|---|
| Named after | Conrad Willgerodt Karl Kindler |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| Organic Chemistry Portal | willgerodt-kindler-reaction |
| RSC ontology ID | RXNO:0000186 |
The related Willgerodt–Kindler reaction[6] takes place with elemental sulfur and an amine like morpholine. The initial product is a thioamide for example that of acetophenone[7] which can again be hydrolyzed to the amide. The reaction is named after Karl Kindler

A possible reaction mechanism for the Kindler variation is depicted below:[8]

The first stage of the reaction is basic
tautomerization
.
References
- .
- .
- .
- ISBN 9780471005285.
- )
- .
- ^ Organic Syntheses, Coll. Vol. 9, p.99 (1998); Vol. 74, p.257 (1997). (Article)
- ISBN 0-471-22854-0
