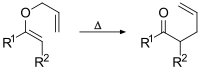Rearrangement reaction
In
Intermolecular rearrangements also take place.
A rearrangement is not well represented by simple and discrete
Three key rearrangement reactions are 1,2-rearrangements, pericyclic reactions and olefin metathesis.
1,2-rearrangements
A 1,2-rearrangement is an organic reaction where a substituent moves from one atom to another atom in a chemical compound. In a 1,2 shift the movement involves two adjacent atoms but moves over larger distances are possible. Skeletal isomerization is not normally encountered in the laboratory, but is the basis of large applications in
Further examples are the Wagner–Meerwein rearrangement:
and the Beckmann rearrangement,[3] which is relevant to the production of certain nylons:[4]
Pericyclic reactions
A pericyclic reaction is a type of reaction with multiple carbon–carbon bond making and breaking wherein the transition state of the molecule has a cyclic geometry, and the reaction progresses in a concerted fashion. Examples are
and the Claisen rearrangement:[5]
Olefin metathesis
Olefin metathesis is a formal exchange of the alkylidene fragments in two alkenes. It is a catalytic reaction with carbene, or more accurately, transition metal carbene complex intermediates.
In this example (ethenolysis, a pair of vinyl compounds form a new symmetrical alkene with expulsion of ethylene.
Other rearragement reactions
1,3-rearrangements
1,3-rearrangements take place over 3 carbon atoms. Examples:
- the Fries rearrangement
- a 1,3-alkyl shift of verbenone to chrysanthenone
See also
- Beckmann rearrangement
- Curtius rearrangement
- Hofmann rearrangement
- Lossen rearrangement
- Schmidt reaction
- Tiemann rearrangement
- Wolff rearrangement
- Photochemical rearrangements
- Thermal rearrangement of aromatic hydrocarbons
- Mumm rearrangement
References
- OCLC 642506595
- ISBN 3-527-30673-0.
- ISBN 978-0-19-927029-3.
- .
- .