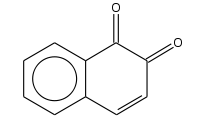
1,2-Naphthoquinone
| |
| Names | |
|---|---|
| Preferred IUPAC name
Naphthalene-1,2-dione | |
| Other names
o-Naphthoquinone,
β-naphthoquinone | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.007.602 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H6O2 | |
| Molar mass | 158.156 g·mol−1 |
| Appearance | yellow solid |
| Melting point | 145 to 147 °C (293 to 297 °F; 418 to 420 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
1,2-Naphthoquinone or ortho-naphthoquinone is a
ferric chloride.[1]
Occurrence
This
diketone (an ortho-quinone) is a metabolite of naphthalene. It arises from the naphthalene-1,2-oxide.[2]
It is also found in diesel exhaust particles. The accumulation of this toxic metabolite in rats from doses of naphthalene has been shown to cause eye damage, including the formation of cataracts.[3]
See also
- 1,4-Naphthoquinone, an isomer of 1,2-naphthoquinone
References
External links
- Troester, M. A.; Lindstrom, A. B.; Waidyanatha, S.; Kupper, L. L.; Rappaport, S. M. (2002). "Stability of Hemoglobin and Albumin Adducts of Naphthalene Oxide, 1,2-Naphthoquinone, and 1,4-Naphthoquinone". PMID 12151627.
- Kikuno, S.; Taguchi, K.; Iwamoto, N.; et al. (2006). "1,2-Naphthoquinone Activates Vanilloid Receptor 1 through Increased Protein Tyrosine Phosphorylation, Leading to Contraction of Guinea Pig Trachea". PMID 16039679.
