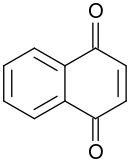
1,4-Naphthoquinone
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Naphthalene-1,4-dione | |
| Other names
1,4-Naphthoquinone
Naphthoquinone α-Naphthoquinone | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.004.526 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H6O2 | |
| Molar mass | 158.15 g/mol |
| Density | 1.422 g/cm3 |
| Melting point | 126 °C (259 °F; 399 K) |
| Boiling point | Begins to sublime at 100 °C |
| 0.09 g/L | |
| -73.5·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
1,4-Naphthoquinone or para-naphthoquinone is a
Preparation
The industrial synthesis involves aerobic oxidation of naphthalene over a vanadium oxide catalyst:[3]
- C10H8 + 3/2 O2 → C10H6O2 + H2O
In the laboratory, naphthoquinone can be produced by the oxidation of a variety of naphthalene compounds. An inexpensive route involves oxidation of naphthalene with chromium trioxide.[4]
Reactions
1,4-Naphthoquinone acts as strong
Uses
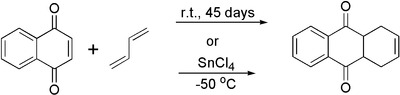
1,4-Naphthoquinone is mainly used as a precursor to anthraquinone by reaction with butadiene followed by oxidation. Nitration gives 5-nitro-1,4-naphthalenedione, precursor to an aminoanthroquinone that is used as a dye precursor.[3]
Derivatives
Naphthoquinone forms the central chemical structure of many natural compounds, most notably the K vitamins. 2-Methyl-1,4-naphthoquinone, called menadione, is a more effective coagulant than vitamin K.
Other natural naphthoquinones include juglone, plumbagin, droserone.
Naphthoquinone
Naphthoquinone functions as a ligand through its electrophilic carbon-carbon double bonds.[7]
Dichlone, a chlorinated derivative of 1,4-naphthoquinone, is used as a fungicide.
See also
References
- ^ Merck Index, 11th Edition, 6315.
- .
- ^ ISBN 978-3527306732.
- ^ Braude, E. A.; Fawcett, J. S. (1953). "1,4-Naphthoquinone" (PDF). Organic Syntheses. 33: 50; Collected Volumes, vol. 4, p. 698.
- PMID 23205621.
- PMID 17867522.
- PMID 15216374.
