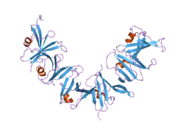AXIN1
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 16: 0.29 – 0.35 Mb | Chr 17: 26.36 – 26.41 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Axin-1 is a protein that in humans is encoded by the AXIN1 gene.[5]
Function
This gene encodes a cytoplasmic protein which contains a regulation of G-protein signaling (RGS) domain and a dishevelled and axin (DIX) domain. The encoded protein interacts with adenomatosis polyposis coli, catenin (cadherin-associated protein) beta 1, glycogen synthase kinase 3 beta, protein phosphatase 2, and itself. This protein functions as a negative regulator of the wingless-type MMTV integration site family, member 1 (WNT) signaling pathway and can induce apoptosis. The crystal structure of a portion of this protein, alone and in a complex with other proteins, has been resolved. Mutations in this gene have been associated with hepatocellular carcinoma, hepatoblastomas, ovarian endometrioid adenocarcinomas, and medulloblastomas. Two transcript variants encoding distinct isoforms have been identified for this gene.[6]
The AXIN proteins attract substantial interest in cancer research as AXIN1 and AXIN2 work synergistically to control pro-oncogenic
Structure
The full-length human protein comprises 862 amino acids with a (predicted) molecular mass of 96 kDa. The N-terminal RGS domain, a GSK3 kinase interacting peptide of Axin1 and homologs of the C-terminal DIX domains have been solved at atomic resolution. Large WNT-downregulating central regions have been characterized as intrinsically disordered by biophysical experiments and bioinformatic analysis.[8] Biophysical destabilization of the folded RGS domain induces formation of nanoaggregates that expose and locally concentrate intrinsically disordered regions, which in turn misregulate Wnt signalling. Many other large IDPs (Intrinsically Disordered Proteins) are affected by missense mutations, such as BRCA1, Adenomatous polyposis coli, CREB-binding protein/(CBP) and might be affected in similar ways by missense mutations of their folded domains.[9]
Interactions
AXIN1 has been shown to
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000103126 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024182 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- S2CID 10565695.
- ^ "Entrez Gene: AXIN1 axin 1".
- PMID 33811251.
- PMID 21087614.
- S2CID 30761541.[permanent dead link]
- ^ S2CID 10875463.
- PMID 12805222.
- ^ PMID 11884395.
- ^ PMID 18632848.
- PMID 10428961.
- ^ PMID 12511557.
- PMID 11336703.
- PMID 10829020.
- PMID 11297546.
- PMID 32965059.
Further reading
- Segditsas S, Tomlinson I (2007). "Colorectal cancer and genetic alterations in the Wnt pathway". Oncogene. 25 (57): 7531–7. S2CID 19546579.
- Flint J, Thomas K, Micklem G, Raynham H, Clark K, Doggett NA, King A, Higgs DR (1997). "The relationship between chromosome structure and function at a human telomeric region". Nat. Genet. 15 (3): 252–7. S2CID 20630444.
- Ikeda S, Kishida S, Yamamoto H, Murai H, Koyama S, Kikuchi A (1998). "Axin, a negative regulator of the Wnt signaling pathway, forms a complex with GSK-3beta and beta-catenin and promotes GSK-3beta-dependent phosphorylation of beta-catenin". EMBO J. 17 (5): 1371–84. PMID 9482734.
- Hart MJ, de los Santos R, Albert IN, Rubinfeld B, Polakis P (1998). "Downregulation of beta-catenin by human Axin and its association with the APC tumor suppressor, beta-catenin and GSK3 beta". Curr. Biol. 8 (10): 573–81. S2CID 2863360.
- Nakamura T, Hamada F, Ishidate T, Anai K, Kawahara K, Toyoshima K, Akiyama T (1998). "Axin, an inhibitor of the Wnt signalling pathway, interacts with beta-catenin, GSK-3beta and APC and reduces the beta-catenin level". Genes Cells. 3 (6): 395–403. S2CID 10875463.
- Hsu W, Zeng L, Costantini F (1999). "Identification of a domain of Axin that binds to the serine/threonine protein phosphatase 2A and a self-binding domain". J. Biol. Chem. 274 (6): 3439–45. PMID 9920888.
- Kitagawa M, Hatakeyama S, Shirane M, Matsumoto M, Ishida N, Hattori K, Nakamichi I, Kikuchi A, Nakayama K, Nakayama K (1999). "An F-box protein, FWD1, mediates ubiquitin-dependent proteolysis of beta-catenin". EMBO J. 18 (9): 2401–10. PMID 10228155.
- Fagotto F, Jho Eh, Zeng L, Kurth T, Joos T, Kaufmann C, Costantini F (1999). "Domains of axin involved in protein-protein interactions, Wnt pathway inhibition, and intracellular localization". J. Cell Biol. 145 (4): 741–56. PMID 10330403.
- Kodama S, Ikeda S, Asahara T, Kishida M, Kikuchi A (1999). "Axin directly interacts with plakoglobin and regulates its stability". J. Biol. Chem. 274 (39): 27682–8. PMID 10488109.
- Jho Eh, Lomvardas S, Costantini F (2000). "A GSK3beta phosphorylation site in axin modulates interaction with beta-catenin and Tcf-mediated gene expression". Biochem. Biophys. Res. Commun. 266 (1): 28–35. PMID 10581160.
- Ikeda S, Kishida M, Matsuura Y, Usui H, Kikuchi A (2000). "GSK-3beta-dependent phosphorylation of adenomatous polyposis coli gene product can be modulated by beta-catenin and protein phosphatase 2A complexed with Axin". Oncogene. 19 (4): 537–45. PMID 10698523.
- Satoh S, Daigo Y, Furukawa Y, Kato T, Miwa N, Nishiwaki T, Kawasoe T, Ishiguro H, Fujita M, Tokino T, Sasaki Y, Imaoka S, Murata M, Shimano T, Yamaoka Y, Nakamura Y (2000). "AXIN1 mutations in hepatocellular carcinomas, and growth suppression in cancer cells by virus-mediated transfer of AXIN1". Nat. Genet. 24 (3): 245–50. S2CID 27531668.
- Spink KE, Polakis P, Weis WI (2000). "Structural basis of the Axin-adenomatous polyposis coli interaction". EMBO J. 19 (10): 2270–9. PMID 10811618.
- Zhang Y, Neo SY, Han J, Lin SC (2000). "Dimerization choices control the ability of axin and dishevelled to activate c-Jun N-terminal kinase/stress-activated protein kinase". J. Biol. Chem. 275 (32): 25008–14. PMID 10829020.
- Daniels RJ, Peden JF, Lloyd C, Horsley SW, Clark K, Tufarelli C, Kearney L, Buckle VJ, Doggett NA, Flint J, Higgs DR (2001). "Sequence, structure and pathology of the fully annotated terminal 2 Mb of the short arm of human chromosome 16". Hum. Mol. Genet. 10 (4): 339–52. PMID 11157797.
- Yamamoto H, Hinoi T, Michiue T, Fukui A, Usui H, Janssens V, Van Hoof C, Goris J, Asashima M, Kikuchi A (2001). "Inhibition of the Wnt signaling pathway by the PR61 subunit of protein phosphatase 2A". J. Biol. Chem. 276 (29): 26875–82. PMID 11297546.
- Mao J, Wang J, Liu B, Pan W, Farr GH, Flynn C, Yuan H, Takada S, Kimelman D, Li L, Wu D (2001). "Low-density lipoprotein receptor-related protein-5 binds to Axin and regulates the canonical Wnt signaling pathway". Mol. Cell. 7 (4): 801–9. PMID 11336703.
- Furuhashi M, Yagi K, Yamamoto H, Furukawa Y, Shimada S, Nakamura Y, Kikuchi A, Miyazono K, Kato M (2001). "Axin facilitates Smad3 activation in the transforming growth factor beta signaling pathway". Mol. Cell. Biol. 21 (15): 5132–41. PMID 11438668.
- Rubinfeld B, Tice DA, Polakis P (2001). "Axin-dependent phosphorylation of the adenomatous polyposis coli protein mediated by casein kinase 1epsilon". J. Biol. Chem. 276 (42): 39037–45. PMID 11487578.