Adenomatous polyposis coli
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) |
|
| |||||||
| Location (UCSC) | n/a | Chr 18: 34.22 – 34.32 Mb | |||||||
| PubMed search | [2] | [3] | |||||||
| View/Edit Human | View/Edit Mouse |
Adenomatous polyposis coli (APC) also known as deleted in polyposis 2.5 (DP2.5) is a
APC is classified as a
The human APC gene is located on the long (q) arm of
Structure
The full-length human protein comprises 2,843 amino acids with a (predicted) molecular mass of 311646 Da. Several N-terminal domains have been structurally elucidated in unique atomistic high-resolution complex structures. Most of the protein is predicted to be intrinsically disordered. It is not known if this large predicted unstructured region from amino acid 800 to 2843 persists in vivo or would form stabilised complexes – possibly with yet unidentified interacting proteins.[9] Recently, it has been experimentally confirmed that the mutation cluster region around the center of APC is intrinsically disordered in vitro.[10]
Role in cancer
The most common mutation in colon cancer is inactivation of APC. In absence of APC inactivating mutations, colon cancers commonly carry activating mutations in
Another mutation is carried by approximately 6 percent[
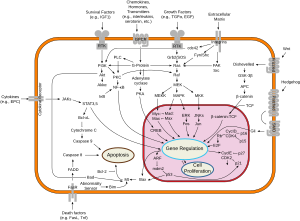
Regulation of proliferation
The (Adenomatous Polyposis Coli) APC protein normally builds a "destruction complex" with glycogen synthase kinase 3-alpha and or beta (
The ability of APC to bind β-catenin has been classically considered to be an integral part of the protein's mechanistic function in the destruction complex, along with binding to Axin through the SAMP repeats.[20] These models have been substantiated by observations that common APC loss of function mutations in the mutation cluster region often remove several β-catenin binding sites and SAMP repeats. However, recent evidence from Yamulla and colleagues have directly tested those models and imply that APC's core mechanistic functions may not require direct binding to β-catenin, but necessitate interactions with Axin.[21] The researchers hypothesized that APC's many β-catenin binding sites increase the protein's efficiency at destroying β-catenin, yet are not absolutely necessary for the protein's mechanistic function. Further research is clearly necessary to elucidate the precise mechanistic function of APC in the destruction complex.
Mutations

Mutations in APC often occur early on in cancers such as colon cancer.[9] Patients with familial adenomatous polyposis (FAP) have germline mutations, with 95% being nonsense/frameshift mutations leading to premature stop codons. 33% of mutations occur between amino acids 1061–1309. In somatic mutations, over 60% occur within a mutation cluster region (1286–1513), causing loss of axin-binding sites in all but one of the 20AA repeats. Mutations in APC lead to loss of β-catenin regulation, altered cell migration and chromosome instability.[11]
Neurological role
Rosenberg et al. found that APC directs cholinergic synapse assembly between neurons, a finding with implications for autonomic neuropathies, for Alzheimer's disease, for age-related hearing loss, and for some forms of epilepsy and schizophrenia.[22] (29)
Interactions
APC (gene) has been shown to
See also
References
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000005871 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 1651563.
- ^ PMID 20018966.
- ^ PMID 28261640.
- PMID 25578398.
- ^ "OrthoMaM phylogenetic marker: APC coding sequence".[permanent dead link]
- ^ PMID 21859464.
- PMID 24130866.
- ^ S2CID 225058221.
- ^ "Familial Adenomatous Polyposis". The Lecturio Medical Concept Library. Retrieved 22 July 2021.
- PMID 23576677.
- ^ PMID 36497357. 5875.
- S2CID 84899068.
- PMID 9556553.
- ^ PMID 9734785.
- PMID 19287945.
- S2CID 29214110.
- PMID 23169527.
- PMID 24931405.
- PMID 20720115.
- PMID 10947987.
- S2CID 12645435.
- ^ PMID 8259519.
- PMID 11712088.
- PMID 12628243.
- PMID 11251183.
- S2CID 664553.
- ^ PMID 11972058.
- PMID 9286858.
- PMID 11707392.
- ^ PMID 7651399.
- PMID 9188857.
- S2CID 10745049.
- PMID 7606712.
- S2CID 14122895.
- PMID 8074697.
- PMID 11389840.
- PMID 15331612.
- S2CID 15004529.
- PMID 12070164.
Further reading
- Cohen MM (November 2003). "Molecular dimensions of gastrointestinal tumors: some thoughts for digestion". American Journal of Medical Genetics. Part A. 122A (4): 303–314. S2CID 9546199.
- Fearnhead NS, Britton MP, Bodmer WF (April 2001). "The ABC of APC". Human Molecular Genetics. 10 (7): 721–733. PMID 11257105.
- Fodde R (May 2002). "The APC gene in colorectal cancer". European Journal of Cancer. 38 (7): 867–871. PMID 11978510.
- Goss KH, Groden J (May 2000). "Biology of the adenomatous polyposis coli tumor suppressor". Journal of Clinical Oncology. 18 (9): 1967–1979. PMID 10784639.
- Järvinen HJ, Peltomäki P (January 2004). "The complex genotype-phenotype relationship in familial adenomatous polyposis". European Journal of Gastroenterology & Hepatology. 16 (1): 5–8. S2CID 20780391.
- Lal G, Gallinger S (June 2000). "Familial adenomatous polyposis". Seminars in Surgical Oncology. 18 (4): 314–323. PMID 10805953.
- van Es JH, Giles RH, Clevers HC (March 2001). "The many faces of the tumor suppressor gene APC". Experimental Cell Research. 264 (1): 126–134. PMID 11237529.
- Rosenberg MM, Yang F, Giovanni M, Mohn JL, Temburni MK, Jacob MH (June 2008). "Adenomatous polyposis coli plays a key role, in vivo, in coordinating assembly of the neuronal nicotinic postsynaptic complex". Molecular and Cellular Neurosciences. 38 (2): 138–152. PMID 18407517.
External links
- GeneReviews/NCBI/NIH/UW entry on APC-Associated Polyposis Conditions
- OMIM entries on APC-Associated Polyposis Conditions
- Adenomatous+Polyposis+Coli+Protein at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- GeneCard
- Database concerning peer-reviewed reports on cancer critical alteration in several genes including (APC (protein)), (TP53), (Beta-catenin|β-catenin)
- Human APC genome location and APC gene details page in the UCSC Genome Browser.




