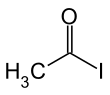
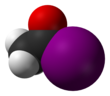
Acetyl iodide
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Acetyl iodide[1] | |||
| Systematic IUPAC name
Ethanoyl iodide | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
ECHA InfoCard
|
100.007.330 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| UN number | 1898 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H3IO | |||
| Molar mass | 169.949 g·mol−1 | ||
| Boiling point | 108 °C; 226 °F; 381 K | ||
| Decomposes | |||
| Thermochemistry | |||
Std enthalpy of (ΔfH⦵298)formation |
-163.18--161.42 kJ mol−1 | ||
| Related compounds | |||
Related acyl halides
|
Acetyl chloride | ||
Related compounds
|
Acetic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Acetyl iodide is an
methyl iodide in the Cativa and Monsanto processes, which are the main industrial processes that generate acetic acid.[2] It is also an intermediate in the production of acetic anhydride from methyl acetate.[3]
Upon treatment with carboxylic acids, acetyl iodide does not exhibit reactions typical of
acyl halides, such as acetyl chloride. Instead, acetyl iodide undergoes iodide/hydroxide exchange with most carboxylic acids:[4]
- CH3COI + RCO2H → CH3CO2H + RCOI
References
- ^ "ACETYL IODIDE - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information.
- Platinum Metals Rev. 44 (3): 94–105. Archived from the original(PDF) on 2015-09-24. Retrieved 2011-07-03.
- S2CID 97920158.