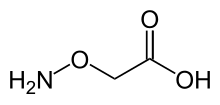
Aminooxyacetic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
(Aminooxy)acetic acid | |
| Other names
Carboxymethoxylamine
Hydroxylamineacetic acid U-7524 | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
IUPHAR/BPS |
|
| MeSH | Aminooxyacetic+Acid |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H5NO3 | |
| Molar mass | 91.066 |
| Density | 1.375 g/cm3 |
| Melting point | 138 °C (280 °F; 411 K) |
| Boiling point | 326.7 °C (620.1 °F; 599.8 K) |
| Hazards | |
| Flash point | 151 °C (304 °F; 424 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aminooxyacetic acid, often abbreviated AOA or AOAA, is a compound that inhibits
Aminooxyacetic acid is a general inhibitor of pyridoxal phosphate (PLP)-dependent enzymes (this includes GABA-T).[3] It functions as an inhibitor by attacking the Schiff base linkage between PLP and the enzyme, forming oxime type complexes.[3]
Aminooxyacetic acid inhibits
Moreover, selective inhibition of aspartate aminotransferase with aminooxyacetic acid ameliorated experimental
Aminooxyacetic acid has been studied as a treatment for
Aminooxyacetic acid also has anticonvulsant properties.[12] At high dosages, it can act as a convulsant agent in mice and rats.[13]
Aminooxyacetic acid can also inhibit
History
Aminooxyacetic acid was first described by Werner in 1893, and was prepared by the hydrolysis of ethylbenzhydroximinoacetic acid.[15][16][17][18] In 1936, Anchel and Shoenheimer used aminooxyacetic acid to isolate ketones from natural sources.[17] Also in 1936, Kitagawa and Takani described the preparation of aminooxyacetic acid by the condensation of benzhydroxamic acid and ethyl bromoacetate, followed by hydrolysis by hydrochloric acid.[19]
