Alfred Werner
Alfred Werner | |
|---|---|
 Werner circa 1915 | |
| Born | 12 December 1866 |
| Died | 15 November 1919 (aged 52) Zürich, Switzerland |
| Nationality | Swiss (from 1895) French |
| Alma mater | Nobel Prize for Chemistry (1913) |
| Scientific career | |
| Fields | Inorganic chemistry |
| Institutions | University of Zurich |
| Doctoral advisor | Arthur Rudolf Hantzsch, Marcellin Berthelot[citation needed] |
Alfred Werner (12 December 1866 – 15 November 1919) was a Swiss
Biography
Werner was born in 1866 in
In his last year, he suffered from a general, progressive, degenerative arteriosclerosis, especially of the brain, aggravated by years of excessive drinking and overwork. He died in a psychiatric hospital in Zürich.[3]
Werner died on 15 November 1919 of arteriosclerosis in Zürich at the age of 52.
Research
Coordination chemistry
In 1893, Werner was the first to propose correct structures for coordination compounds containing
For example, it was known that cobalt forms a "complex" hexamine cobalt (III) chloride, with formula CoCl3•6NH3, but the nature of the association indicated by the dot was mysterious. Werner proposed the structure

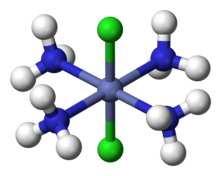
For complexes with more than one type of ligand, Werner succeeded in explaining the number of
Werner also prepared complexes with
Nature of valence
Before Werner, chemists defined the valence of an element as the number of its bonds without distinguishing different types of bonds. However, in complexes such as [Co(NH3)6]Cl3 for example, Werner considered that the Co-Cl bonds correspond to a "primary" valence of 3 at long distance, while the Co-NH3 bonds which correspond to a "secondary" or weaker valence of 6 at shorter length. This secondary valence of 6 he referred to as the coordination number which he defined as the number of molecules (here of NH3) directly linked to the central metal atom. In other complexes, he found coordination numbers of 4 or 8.
On these views, and other similar views, in 1904 Richard Abegg formulated what is now known as Abegg's rule which states that the difference between the maximum positive and negative valence of an element is frequently eight. This rule was used later in 1916 when Gilbert N. Lewis formulated the "octet rule" in his cubical atom theory.
In modern terminology, Werner's primary valence corresponds to the
Works
- Lehrbuch der Stereochemie . Fischer, Jena 1904 Digital edition by the University and State Library Düsseldorf
References
- ^ "Alfred Werner - Nobel Prize for Chemistry 1913" (PDF). University of Zurich. Retrieved 9 December 2022.
He moved there with his wife, Emma Wilhelmine, née Giesker, whom he had married on 1 October 1894.
- ^ https://www.nobelprize.org/nobel_prizes/chemistry/laureates/1913/werner-bio.html Nobel Prize Retrieved 1 December 2012
- ^ a b c d e "Alfred Werner - Swiss chemist". britannica.com. Retrieved 14 April 2018.
External links
- Alfred Werner on Nobelprize.org including the Nobel Lecture, 11 December 1913 On the Constitution and Configuration of Higher-Order Compounds
- The Nobel Prize in Chemistry 1913 - short article about his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research, especially in inorganic chemistry.

