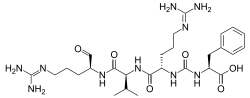
Antipain
| |
| Names | |
|---|---|
| IUPAC name
N2-{[(1S)-1-carboxy-2-phenylethyl]carbamoyl}-N5-(diaminomethylidene)-L-ornithyl-N-{(2S)-5-[(diaminomethylidene)amino]-1-oxopentan-2-yl}-L-valinamide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C27H44N10O6 | |
| Molar mass | 604.713 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Antipain is an
It has been crystallized in complexes with carboxypeptidase, which is obtained from wheat,[4] and Leishmania major oligopeptidase B.[5] In both cases, the backbone carbonyl of the terminal arginine of antipain forms a covalent bond to the active site serine in the protease.
A study was performed for information on the effect of antipain on the quality of post-thawed ram semen.[6] The results from this experiment concluded that antipain aided in the quality of ram semen by maintaining the sperm mobility.[6] Antipain includes the function to inhibit a degrading enzyme, called plasmin, permitting this substance to be able to improve the resistance of membrane disruption by freezing temperatures.[6]
Antipain Y, a similar chemical compound that is an
