Aporphine alkaloids

Aporphine alkaloids are naturally occurring chemical compounds from the group of alkaloids. After the benzylisoquinoline alkaloids they are the second largest group of isoquinoline alkaloids.
At least 85 aporphine alkaloids have been isolated from plants of 15 families. The best known representative is apomorphine. The aporphine alkaloids are of interest mainly because of their similarity to morphine.
Occurrence

The aporphine alkaloids are most commonly found in plants. For example,
was first found in the horn poppy and usually the name of the alkaloids is derived from the plants in which they were first found.Corydin as a further representative of the aporphine alkaloids is found in Corydalis (larkspurs) Dicentra (heart flowers), and also in the horn poppy.
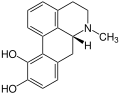
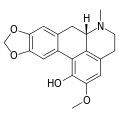
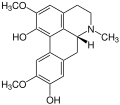
Examples
-
Isoboldine
The aporphine alkaloids differ in their substituents and their position on the base structure. Furthermore, their stereochemistry is partly different; most often they are (R)-configured, but glaucine, bulbocapnine, and isothebaine, for example, are (S)-configured.[1]
Biosynthesis
The method by which the central aporphine ring structure is constructed in nature is exemplified by the
Chemistry

The aporphine alkaloids are of particular interest because of their proximity to morphine and benzylisoquinoline alkaloids. For example, as the name suggests, morphine can be used to produce apomorphine. This can be done by adding an acid under the influence of heat.
The proaporphin alkaloids and the aporphin alkaloids share a framework isomerism.
The aporphine alkaloids usually have a stereocenter.
The (R)-configured glaucine can be synthesized from (S)-glaucine.
Uses

Cassytha filiformis, a plant used in African traditional medicine, contains many aporphine alkaloids and that the three main alkaloids actinodaphnin, cassythin, and dicentrin have an in vitro effect on cancer cells.[4]
References
- .
- S2CID 85841935.
- ISBN 0-471-03478-9.
- PMID 15124084.