Dehydration reaction
In chemistry, a dehydration reaction is a chemical reaction that involves the loss of water from the reacting molecule or ion. Dehydration reactions are common processes, the reverse of a hydration reaction.
Dehydration reactions in organic chemistry
Esterification
The classic example of a dehydration reaction is the
- RCO2H + R′OH ⇌ RCO2R′ + H2O
Often such reactions require the presence of a dehydrating agent, i.e. a substance that reacts with water.
Etherification
Two
Nitrile formation
Nitriles are often prepared by dehydration of primary amides.
- RC(O)NH2 → RCN + H2O
Ketene formation
Ketene is produced by heating acetic acid and trapping the product:[1]
- CH3CO2H → CH2=C=O + H2O
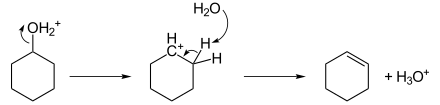
Alkene formation
Alkenes can be made from alcohols by dehydration. This conversion, among others, is used in converting biomass to liquid fuels.[2] The conversion of ethanol to ethylene is a fundamental example:[3][4]
- CH3CH2OH → H2C=CH2 + H2O
The reaction is accelerated by
Some alcohols are prone to dehydration. 3-Hydroxylcarbonyls, called aldols, release water upon standing at room temperature:
- RC(O)CH2CH(OH)R' → RC(O)CH=CHR' + H2O
The reaction is induced by dehydrating reagents. For example, 2-methyl-cyclohexan-1-ol dehydrates to 1-methylcyclohexene in the presence of Martin's sulfurane, which reacts irreversibly with water.[6][7]
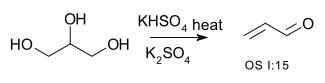
Double dehydration is illustrated by the conversion of glycerol to acrolein:[8][9]
Dehydration reactions in inorganic chemistry
The formation of the pyrophosphate bond is an important dehydration relevant to bioenergetics.
Various construction materials are produced by dehydration.
- heat (released as steam).
The resulting dry powder is ready to be mixed with water to form a stiff but workable paste that hardens.
See also
References
- ISBN 978-3527306732.
- PMID 24083630.
- ISBN 978-3527306732.
- ISSN 0888-5885.
- .
- .
- ISBN 0471936235.
- .
- ISSN 1463-9262.
- ^ Staff. "CaSO4, ½ H2O". LaFargePrestia. Archived from the original on November 20, 2008. Retrieved 27 November 2008.