Atmosphere of Mercury
Molecular oxygen < 9 × 1014; < 2.5 × 107 | | |
| Sodium | ~ 2 × 1011; 1.7–3.8 × 104 | |
|---|---|---|
| Potassium | ~ 2 × 109; ~ 4000 | |
| Calcium | ~ 1.1 × 108; ~ 3000 | |
| Magnesium | ~ 4 × 1010; ~ 7.5 × 103 | |
| Argon | ~ 1.3 × 109; < 6.6 × 106 | |
| Water | < 1 × 1012; < 1.5 × 107 | |
Mercury, being the closest to the Sun, with a weak magnetic field and the smallest mass of the recognized terrestrial planets, has a very tenuous and highly variable atmosphere (surface-bound exosphere) containing hydrogen, helium, oxygen, sodium, calcium, potassium and water vapor, with a combined pressure level of about 10−14 bar (1 nPa).[2] The exospheric species originate either from the Solar wind or from the planetary crust. Solar light pushes the atmospheric gases away from the Sun, creating a comet-like tail behind the planet.
The existence of a Mercurian atmosphere was contentious until 1974, although by that time a consensus had formed that Mercury, like the Moon, lacked any substantial atmosphere. This conclusion was confirmed in 1974 when the unmanned Mariner 10 spaceprobe discovered only a tenuous exosphere. Later, in 2008, improved measurements were obtained by the MESSENGER spacecraft, which discovered magnesium in the Mercurian exosphere.
Composition
The Mercurian exosphere consists of a variety of species originating either from the
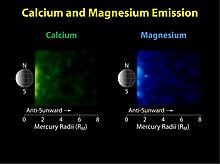
The fourth species detected in Mercury's exosphere was
In 2008 the
Properties
Mariner 10's ultraviolet observations have established an upper bound on the exospheric surface density at about 105 particles per cubic centimeter. This corresponds to a surface pressure of less than 10−14 bar (1 nPa).[14]
The temperature of Mercury's exosphere depends on species as well as geographical location. For exospheric atomic hydrogen, the temperature appears to be about 420 K, a value obtained by both Mariner 10 and MESSENGER.[4] The temperature for sodium is much higher, reaching 750–1,500 K on the equator and 1,500–3,500 K at the poles.[15] Some observations show that Mercury is surrounded by a hot corona of calcium atoms with temperature between 12,000 and 20,000 K.[9] In the early 2000s, a simulation of Mercury's Na exosphere and its temporal variation was conducted to identify the source process that supplied crustal species to the exosphere. Processes like; evaporation, diffusion from the interior, sputtering by photons and energetic ions, chemical sputtering by photons, and meteoritic vaporization were tested. However, evaporation provides the strongest match when comparing the changes in the sodium exosphere with solar distance and time of day to the 2001 observations of Mercury's sodium tail.[16]
Tails


Because of Mercury's proximity to the Sun, the pressure of solar light is much stronger than near Earth. Solar radiation pushes neutral atoms away from Mercury, creating a comet-like tail behind it.[17] The main component in the tail is sodium, which has been detected beyond 24 million km (1000 RM) from the planet.[18] This sodium tail expands rapidly to a diameter of about 20,000 km at a distance of 17,500 km.[19] In 2009, MESSENGER also detected calcium and magnesium in the tail, although these elements were only observed at distances less than 8 RM.[17]
Observation difficulties
Mercury is the least explored planet of the
Instead, flyby and orbital missions to Mercury can study the planet and receive accurate data. Even though Mercury is closer to Earth than Pluto is, the transfer orbit from Earth to Mercury requires more energy. Mercury being so close to the Sun, space probes going there are accelerating as they approach, due to the Sun's gravitational pull. This requires the use of retrorockets, which use fuel that the probe must carry instead of better instruments.[20]
See also
- Orders of magnitude (pressure)
- Magnetosphere of Mercury
References
Notes
- ^ Killen 2007, p. 456, Table 5
- ^ "NASA—Mercury". Archived from the original on 2005-01-05. Retrieved 2009-09-26.
- ^ a b c Killen, 2007, pp. 433–434
- ^ a b McClintock 2008, p. 93
- ^ a b Killen, 2007, pp. 434–436
- ^ Killen, 2007, pp. 438–442
- ^ Killen, 2007, pp. 442–444
- ^ Killen, 2007, pp. 449–452
- ^ a b Killen, 2007, pp. 452–453
- ^ a b McClintock 2009, p. 612–613
- .
- ^ "MESSENGER Scientists 'Astonished' to Find Water in Mercury's Thin Atmosphere". The Planetary Society. 2008-07-03. Archived from the original on 6 April 2010. Retrieved 2010-03-28.
- ^ Zurbuchen 2008, p. 91, Table 1
- ^ Domingue, 2007, pp. 162–163
- ^ Killen, 2007, pp. 436–438
- ^ .
- ^ a b McClintock 2009, p. 610–611
- ^ Schmidt 2010, p. 9–16
- ^ Killen, 2007, p. 448
- .
Bibliography
- Domingue, Deborah L.; Koehn, Patrick L.; Killen, Rosemary M.; et al. (2007). "Mercury's Atmosphere: A Surface-Bounded Exosphere". S2CID 121301247.
- Fink, Uwe; Larson, Harold P.; Poppen, Richard F. (1974). "A new upper limit for an atmosphere of CO2, CO on Mercury". doi:10.1086/180075.
- Killen, Rosemary; Cremonese, Gabrielle; Lammer, Helmut; et al. (2007). "Processes that Promote and Deplete the Exosphere of Mercury". S2CID 121944553.
- McClintock, William E.; Bradley, E. Todd; Vervack Jr, Ronald J.; et al. (2008). "Mercury's Exosphere: Observations During MESSENGER's First Mercury Flyby". Science. 321 (5885): 92–94. S2CID 6857425.
- Schmidt, Carl A.; Wilson, Jody K.; Baumgardner, Jeff; Mendillo, Michael (2010). "Orbital effects on Mercury's escaping sodium exosphere". Icarus. 207 (1): 9–16. .
- McClintock, William E.; Vervack Jr, Ronald J.; Bradley, E. Todd; et al. (2009). "MESSENGER Observations of Mercury's Exosphere: Detection of Magnesium and Distribution of Constituents". Science. 324 (5927): 610–613. S2CID 5578520.
- Rasool, S.I.; Gross, S.H.; McGovern, W.E. (1966). "The atmosphere of Mercury". S2CID 120501658.
- Williams, I.P. (1974). "Atmosphere of Mercury". S2CID 4198611.
- Zurbuchen, Thomas H.; Raines, Jim M.; Gloeckler, George; et al. (2008). "MESSENGER Observations of the Composition of Mercury's Ionized Exosphere and Plasma Environment". Science. 321 (5885): 90–92. S2CID 206513512.
