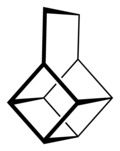
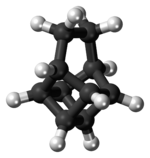
Basketane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Pentacyclo[4.4.0.02,5.03,8.04,7]decane | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C10H12 | |||
| Molar mass | 132.206 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Basketane is a polycyclic alkane with the chemical formula C10H12. The name is taken from its structural similarity to a basket shape. Basketane was first synthesized in 1966, independently[1] by Masamune[2] and Dauben and Whalen.[3] A patent application published in 1988 used basketane, which is a hydrocarbon, as a source material in doping thin diamond layers because of the molecule's high vapor pressure, carbon ring structure, and fewer hydrogen-to-carbon bond ratio.[4]
Chemical nomenclature
In the year 1989 and before the synthesis of basketane, historic chemists were intrigued by the structural make-up of molecules, specifically those in objects seen in everyday life.[5] Using supramolecular chemistry, molecules such as cubane and basketane were named according to their corresponding shape and historically revealed certain characteristics and personal motives of chemists at that time.[5] Naming these uniquely shaped molecules were also done considering chemical nomenclature such as adding "-anes" for single carbon-carbon bonds and "-enes" for double carbon-carbon bonds to the end of the appropriate molecules.[6]
Synthesis
One synthesis of basketane begins with a Diels–Alder reaction between cyclooctatetraene (1) and maleic anhydride (2), giving the polycyclic anhydride 3, which photoisomerizes in acetone via an intramolecular cyclization to give 4 at a 40% yield. Hydrolysis of the anhydride followed by treatment with lead tetraacetate affords the unsaturated basketene (5), which is then hydrogenated to basketane (6).[2]
An alternative synthetic route with better overall yield uses 1,4-benzoquinone and cyclohexa-1,3-diene as starting materials. 1,4-Benzoquinone (1) is first converted to 2,5-dibromo-1,4-benzoquinone (2), which reacts in a Diels–Alder reaction with cyclohexa-1,3-diene (3) to form the polycyclic diketone 4. This diketone photoisomerizes to 1,6-dibromopentacyclo[6.4.0.03,6.04,12.05,9]dodeca-2,7-dione (5), which undergoes a pseudo-Favorskii rearrangement in a 25% aqueous solution of sodium hydroxide, giving the dicarboxylic acid 6. The acid is decarboxylated with a modified Hunsdiecker reaction to a dibromide 7, which is reductively debrominated with tributyltin hydride to basketane (8) at a 11% yield relative to the starting material cyclohexa-1,3-diene.[7]
A 1994 synthesis starts with homocubanone, a cubane derivative, forming basketane via the basketyl
Properties and reactions
The C-C-C and H-C-C bond angles in basketane and several other cages deviate from 109.5 degrees, i.e., they are
Metal-catalyzed rearrangements
Transition metals catalyze the
Other transition metals catalyze the formation of a snoutane derivative with a tricyclic diene. The ratio of the products depends on the nature of the catalyst used and the substitutions on the basketane.[12]
Basketane absorbs an equivalent amount of hydrogen gas in the presence of palladium on carbon, After some initial confusion, it was shown that the C3-C4 bond is hydrogenolyzed to give the dihydrobasketane tetracyclo[4.4.0.02,5.03,8]decane.[13] Further hydrogenation cleaves the C5-C6 bond to give the hydrocarbon twistane.[14]
See also
References
Further reading
- Binmore, Gavin T.; Della, Ernest W.; Elsey, G. M.; Head, N. J.; Walton, J. C. (1994). "Homolytic Reactions of Homocubane and Basketane: Rearrangement of the 9-Basketyl Radical by Multiple β-Scissions". .