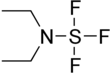Diethylaminosulfur trifluoride
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
N,N-Diethyl-S,S,S-trifluoro-λ4-sulfanamine | |||
| Other names
diethyl(trifluorosulfido)amine
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| Abbreviations | DAST | ||
| ChemSpider | |||
ECHA InfoCard
|
100.048.866 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H10F3NS | |||
| Molar mass | 161.19 g·mol−1 | ||
| Appearance | colourless oil | ||
| Density | 1.220 g/cm3 | ||
| Boiling point | 30 to 32 °C (86 to 90 °F; 303 to 305 K) at 3 mmHg | ||
| Reacts with water | |||
| Solubility | reacts with ethanol soluble[vague] in acetonitrile | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
corrosive, flammable, can be explosive | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H226, H302, H312, H314, H332 | |||
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P280, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P363, P370+P378, P403+P235, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Diethylaminosulfur trifluoride (DAST) is the
Use in organic synthesis
DAST converts
Synthesis
DAST is prepared by the reaction of diethylaminotrimethylsilane and sulfur tetrafluoride:[3]
- Et2NSiMe3 + SF4 → Et2NSF3 + Me3SiF
The original paper calls for trichlorofluoromethane (Freon-11) as a solvent.[4] Diethyl ether is a green alternative that can be used with no decrease in yield.[5] Because of the dangers involved in the preparation of DAST (glass etching, possibility of exothermic events), it is often purchased from a commercial source. At one time Carbolabs[6] was one of the few suppliers of the chemical but a number of companies now sell DAST. Carbolabs was acquired by Sigma-Aldrich in 1998.
Safety and alternative reagents
Upon heating, DAST converts to the highly explosive (NEt2)2SF2 with expulsion of sulfur tetrafluoride. To minimize accidents, samples are maintained below 50 °C.[7] Bis-(2-methoxyethyl)aminosulfur trifluoride (trade name: Deoxo-Fluor) and difluoro(morpholino)sulfonium tetrafluoroborate (trade name: XtalFluor-M) are reagents related to DAST with less explosive potential.[8][9][10] XtalFluor-E has been jointly developed by OmegaChem Inc. and Manchester Organics Ltd. in 2009–2010.[9][10]
See also
References
- ^ ISBN 0-471-93623-5.
- ^ Markovskii, LN; Pashinnik, VE; KIRSANOVA, NA (1975). "Sulfur Bis(dialkylamino Difluorides". Zhurnal Organicheskoi Khimii. 11: 74–77 – via Web of Science.
- ^ W. J. Middleton, E. M. Bingham "Diethylaminosulfur Trifluoride" Organic Syntheses, Coll. Vol. 6, p.440; Vol. 57, p.50. Online version
- ISSN 0022-3263.
- .
- ^ REACTION OF SULFOXIDES WITH DIETHYLAMINOSULFUR TRIFLUORIDE: FLUOROMETHYL PHENYL SULFONE, A REAGENT FOR THE SYNTHESIS OF FLUOROALKENES, Organic Syntheses, Coll. Vol. 9, p.446 (1998); Vol. 72, p.209 (1995)
- .
- doi:10.1039/a808517j.
- ^ PMID 20405933.
- ^ PMID 19799406.