Endothelial cell tropism
Endothelial cell tropism or endotheliotropism is a type of
Cellular features and mechanisms
There are a multitude of endothelial cell features that influence cell tropism and ultimately, contribute to endothelial cell activation and dysfunction as well as the continuation of the virus life cycle.
Cell surface receptors

Transcription Factors & Viral Replication
After entry into the cell, these intracellular parasites require factors in the host cell to support viral replication and release of progeny virions. Specifically, the host factors include proteins, such as transcription factors and polymerases, which aid in replicating the viral genome.[7] Therefore, the sole entry into a live host does not necessarily result in propagation for viral progeny as the cell may not contain the critical transcription factors or polymerases for virus replication. Furthermore, within the viral genome, there are not only instructions to synthesize viral proteins but also other virulence factors such as genes, cellular structures, and other regulatory processes that enable a pathogen to control the host's antiviral responses.[8] These virulence factors can counter the host defense mechanisms that attempt eliminate the infection via the host's immune system.
Host defense mechanisms
Endothelial cells also possess intrinsic antiviral responses which leverage the host's immune system to battle the infection or restrict viral replication.
Examples and effects on viral pathogenesis

Flaviviruses
Dengue is an example of a mosquito-borne flavivirus that causes Dengue fever. While endothelial cells are not the major cell type Dengue targets, the virus binds to various cell surface receptors on endothelial cells with particular productive infection via heparan sulfate-containing cell surface receptors.[14] The infection of the endothelium via these receptors have been indicated to impair critical immune responses and alter capillary permeability which in turn support the clinical course of the disease.[15]
Filoviruses

Ebola is one viral hemorrhagic fever virus that causes Ebola Virus Disease (EVD). Analysis of human samples of nonsurvivors of the disease have shown that the endothelium is significantly changed from the healthy state.[16][17] Other alterations from homeostasis include the widespread expression of viral antigens in endothelial cells.[18] The glycoprotein of the virus, which serves as the virus's "key" into the cell, has been indicated to majorly damage the endothelium.[16] For instance, the liver has been highly implicated as an area of damage upon infection. Liver sinusoidal endothelial cells (LSEC) express a variety of scavenger receptors including FcγRIIb2 and mannose receptor which are critical in eliminating waste molecules in the liver but also engulf ligands via the CME pathway.[19] In addition to supporting entry of virus, the interactions to these receptors also may also hinder the clearance of pharmaceuticals given to mitigate the infection.
Orthomyxoviridae
Applications
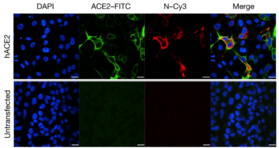
Technologies of study
Depending on the biosafety level (BSL) also known as the pathogen or protection level, there are different levels of biocontainment and approvals required to study the pathogen; this protection level affects how and where the pathogen is studied.[21] While these summarizes focus on endothelial cell tropism, these techniques also apply broadly to various methods in virology. These summaries do not provide comprehensive list but are representative of common platforms to study emerging infectious diseases.
In vitro approaches
Immortalized cells offer a renewable resource to study variety of pathogens. The characterization of the endothelial tropism allows researchers to modify either the cell to display the receptor that the virus's glycoprotein interacts with to attach to the cell. However, these
In vivo models of infection

Nonhuman primates such as rhesus macaques serve as the "gold standard" approach for animal models for many BSL4 pathogens when the biological phenomenon cannot be studied in other species.[23] As many infectious diseases are zoonotic in nature, modeling these diseases in these macaque species which have some biological similarities to humans provide insight into disease understanding in circumstances which a virus is poorly understood and treatment options are limited or nonexistent. The readouts of these models can be evaluated through tissue samples or blood samples, for instance. However, these in vivo models of infection such as rodent and nonhuman primate models have presented ethical concerns and shortcomings as it involves laboratory confinement of an animal and introducing to it a disease insult.[24] The emergence of advanced in vivo including humanized or transgenic rodent models provide an alternative to the macaque series but also harbor concerns if these models recapitulate human physiology or are predictive of human-like responses to a disease or therapeutic. These models involve genetically modifying and/or transplanting human tissue into a rodent model.[25] In conjunction to in vitro cell-based assays, these in vivo models are critical to validate therapeutics during drug discovery and development.
Drug discovery and development
Endothelial cell tropism informs medical countermeasures in response to an emerging infectious diseases. These medical countermeasures include how therapeutics such as small molecules compounds and vaccines are developed.
Antivirals and other small molecule drugs
| Group | Drug(s) | Mechanism of Action | References |
|---|---|---|---|
| Viral Entry Inhibitors | Maraviroc
Inmazeb
|
Blocks receptor engagement, endocytosis/macropinocytosis, attachment, fusion or signaling involved uptake of the virus | [26][29][30] |
| Viral Protein Synthesis Inhibitors | Lopinavir/ritonavir | Suppresses or slows virus replication by disrupting processes involved in translation or generation of protein | [31] |
| Viral Polymerase Inhibitors | Molnupiravir | Interferes with regulation of transcription of viral proteins during viral replication | [32] |
| Immunomodulators | Nitazoxanide | Interfere and counter with host regulated pathways during viral replication (e.g., IFN pathways and mechanism of viral RNA sensing) | [26][33] |
Vaccines
Vaccines are therapeutics that are preventative measure to infectious diseases. These therapeutics offer the body adaptive immunity to a specific pathogen. Fundamentally, vaccines provide patients protection by eliciting an immune response so that they develop antibodies that will help protect against the invading pathogen. The development, production, and global distribution of these vaccines is imperative to prevent, control, and eradicate pandemic potential pathogens. Specifically, cell cultured-based vaccine technologies utilize cell lines that have a wide range of viral tropism to adapt virus strains used in the development of vaccines to new cells.[34] This application of cell tropism evaluates the diverse viral entry pathways and host receptors to accomplish this goal. Moreover, the aspects endothelial cell activation and dysfunction become important readouts during vaccine development as they are part the hallmarks of many clinical courses of infectious diseases. One of the most promising vaccine candidates for Ebola is Merck's recombinant VSV-EBOV vaccine, Ervebo. The vaccine was critical during the end of 2014/2015 Ebola outbreak in Guinea. Ervebo was shown to be effective in nonhuman primate and later in Guinea during the authorized human efficacy trial which showed that Ervebo was also highly protective in humans. The vaccine employs VSV as the surrogate to display the Ebola glycoprotein.[35] VSV does not cause disease in humans which renders it a useful backbone to hold an important protein of Zaire Ebola virus. When the vaccine is administered, the recombinant VSV introduces a functional Ebola virus glycoprotein which interacts with endothelial cell barrier and elicit a rapid immune response without causing disease in patients. Therefore, the development and scaling of vaccines involves important considerations to endothelial cell tropism.
References
- ^ PMID 33718448.
- S2CID 15235357.
- S2CID 4380108.
- PMID 25085912.
- ^ Yuan, Sarah Y.; Rigor, Robert R. (2010). Signaling Mechanisms in the Regulation of Endothelial Permeability. Morgan & Claypool Life Sciences.
- PMID 33375371.
- ISBN 978-0122530555
- S2CID 237387636.
- PMID 19696766.
- PMID 34646033.
- PMID 25301932.
- PMID 33361764.
- PMID 34721846.
- PMID 29430005.
- PMID 18522648.
- ^ PMID 26024394.
- PMID 16051836.
- PMID 33080902.
- PMID 34707514.
- PMID 25520707.
- PMID 30570972, retrieved 2021-11-01
- PMID 28870117.
- PMID 32674252.
- PMID 30597951.
- PMID 33291453.
- ^ PMID 33726557.
- PMID 22312262.
- PMID 25601858.
- PMID 23199399.
- PMID 33432551.
- PMID 34335286.
- PMID 34742052.
- PMID 32574331.
- PMID 32202955.
- PMID 28081225.
