Monoclonal antibody therapy
Major applications include
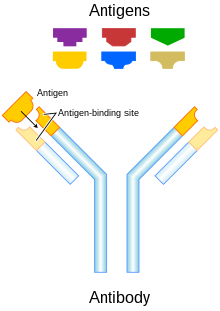
Antibody structure and function
Monoclonal antibody therapy may prove to be beneficial for
History
Four major antibody types that have been developed are
Murine
Initial therapeutic antibodies were murine
Initially, murine antibodies were obtained by hybridoma technology, for which Jerne, Köhler and Milstein received a Nobel prize. However the dissimilarity between murine and human immune systems led to the clinical failure of these antibodies, except in some specific circumstances. Major problems associated with murine antibodies included reduced stimulation of
Chimeric and humanized
To reduce murine antibody
Humanised antibodies are produced by grafting murine hypervariable regions on amino acid domains into human antibodies. This results in a molecule of approximately 95% human origin. Humanised antibodies bind antigen much more weakly than the parent murine monoclonal antibody, with reported decreases in affinity of up to several hundredfold.
Human monoclonal antibodies
Human monoclonal antibodies (suffix -umab) are produced using
The heavy and light chains of human IgG proteins are expressed in structural polymorphic (allotypic) forms. Human IgG allotype is one of the many factors that can contribute to immunogenicity.[16][17]
Targeted conditions
Cancer
Anti-cancer monoclonal antibodies can be targeted against malignant cells by several mechanisms. Ramucirumab is a recombinant human monoclonal antibody and is used in the treatment of advanced malignancies.[18] In childhood lymphoma, phase I and II studies have found a positive effect of using antibody therapy.[19]
Monoclonal antibodies used to boost an anticancer immune response is another strategy to fight cancer where cancer cells are not targeted directly. Strategies include antibodies engineered to block mechanisms which downregulate anticancer immune responses, checkpoints such as PD-1 and CTLA-4 (checkpoint therapy),[20] and antibodies modified to stimulate activation of immune cells.[21]
Autoimmune diseases
Monoclonal antibodies used for
Alzheimer's disease
Alzheimer's disease (AD) is a multi-faceted, age-dependent, progressive neurodegenerative disorder, and is a major cause of dementia.
However, mAbs are large molecules and due to the blood–brain barrier, uptake of mAb into the brain is extremely limited, only approximately 1 of 1000 mAb molecules is estimated to pass.[25] However, the Peripheral Sink hypothesis proposes a mechanism where mAbs may not need to cross the blood–brain barrier.[26] Therefore, many research studies are being conducted from failed attempts to treat AD in the past.[24]
However, anti-Aβ vaccines can promote antibody-mediated clearance of Aβ plaques in transgenic mice models with amyloid precursor proteins (APP), and can reduce cognitive impairments.
Currently, there are two FDA approved antibody therapies for Alzheimer's disease, Aducanemab and Lecanemab. Aducanemab has received accelerated approval while Lecanemab has received full approval.[25] Several clinical trials using passive and active immunization have been performed and some are on the way with expected results in a couple of years.[24][25] The implementation of these drugs is often during the early onset of AD. Other research and drug development for early intervention and AD prevention is ongoing. Examples of important mAb drugs that have been or are under evaluation for treatment of AD include Bapineuzumab, Solanezumab, Gautenerumab, Crenezumab, Aducanemab, Lecanemab and Donanemab.[25]
Bapineuzumab
In Phase III clinical trials, Bapineuzumab showed promising positive effect on biomarkers of AD but failed to show effect on cognitive decline. Therefore, Bapineuzumab was discontinued after failing in the Phase III clinical trial.[29]
Solanezumab
Solanezumab, an anti-Aβ mAb, targets the N-terminus of Aβ. In Phase I and Phase II of clinical trials, Solanezumab treatment resulted in cerebrospinal fluid elevation of Aβ, thereby showing a reduced concentration of Aβ plaques. Additionally, there are no associated adverse side effects. Phase III clinical trials of Solanezumab brought about significant reduction in cognitive impairment in patients with mild AD, but not in patients with severe AD. However, Aβ concentration did not significantly change, along with other AD biomarkers, including phospho-tau expression, and hippocampal volume. Phase III clinical trials of Solanezumab failed as it did not show effect on cognitive decline in comparison to placebo. [30]
Lecanemab
Lecanemab (BAN2401), is a humanized mAb that selectively targets toxic soluble Aβ protofibrils,[31] In phase 3 clinical trials,[32] Lecanemab showed a 27% slower cognitive decline after 18 months of treatment in comparison to placebo.[33][34] The phase 3 clinical trials also reported infusion related reactions, amyloid-related imaging abnormalities and headaches as the most common side effects of Lecanemab. In July 2023 the FDA gave Lecanemab full approval for the treatment of Alzheimer's Disease [35] and it was given the commercial name Leqembi.
Preventive trials
Failure of several drugs in Phase III clinical trials has led to AD prevention and early intervention for onset AD treatment endeavours. Passive anti-Aβ mAb treatment can be used for preventive attempts to modify AD progression before it causes extensive brain damage and symptoms. Trials using mAb treatment for patients positive for genetic risk factors, and elderly patients positive for indicators of AD are underway. This includes anti-AB treatment in Asymptomatic Alzheimer's Disease (A4), the Alzheimer's Prevention Initiative (API), and DIAN-TU.[26] The A4 study on older individuals who are positive for indicators of AD but are negative for genetic risk factors will test Solanezumab in Phase III Clinical Trials, as a follow-up of previous Solanezumab studies.[26] DIAN-TU, launched in December 2012, focuses on young patients positive for genetic mutations that are risks for AD. This study uses Solanezumab and Gautenerumab. Gautenerumab, the first fully human MAB that preferentially interacts with oligomerized Aβ plaques in the brain, caused significant reduction in Aβ concentration in Phase I clinical trials, preventing plaque formation and concentration without altering plasma concentration of the brain. Phase II and III clinical trials are currently being conducted.[26]
Therapy types
Radioimmunotherapy
Antibody-directed enzyme prodrug therapy
Antibody-drug conjugates
Immunoliposome therapy
Immunoliposomes are antibody-conjugated
Checkpoint therapy
Checkpoint therapy uses antibodies and other techniques to circumvent the defenses that tumors use to suppress the immune system. Each defense is known as a checkpoint. Compound therapies combine antibodies to suppress multiple defensive layers. Known checkpoints include
The
FDA-approved therapeutic antibodies
This section needs to be updated. (May 2021) |
The first FDA-approved therapeutic monoclonal antibody was a murine IgG2a CD3 specific
| Antibody | Brand name | Company | Approval date | Route | Type | Target | Indication (Targeted disease) |
BLA STN | Drug Label |
|---|---|---|---|---|---|---|---|---|---|
| abciximab | ReoPro | Centocor |
12/22/1994 | intravenous | chimeric Fab | GPIIb/IIIa |
Percutaneous coronary intervention | 103575 | Link |
| adalimumab | Humira | Abbvie |
12/31/2002 | subcutaneous | fully human | TNF |
Rheumatoid arthritis | 125057 | Link |
adalimumab-atto |
Amjevita | Amgen | 9/23/2016 | subcutaneous | fully human, biosimilar | TNF |
Plaque psoriasis |
761024 | Link |
ado-trastuzumab emtansine |
Kadcyla | Genentech | 2/22/2013 | intravenous | humanized, antibody-drug conjugate |
HER2 | Metastatic breast cancer | 125427 | Link |
| alemtuzumab | Campath, Lemtrada | Genzyme | 5/7/2001 | intravenous | humanized | CD52 | B-cell chronic lymphocytic leukemia |
103948 | Link |
| alirocumab | Praluent | Sanofi Aventis |
7/24/2015 | subcutaneous | fully human | PCSK9 | Heterozygous familial hypercholesterolemia Refractory hypercholesterolemia |
125559 | Link |
| atezolizumab | Tecentriq | Genentech | 5/18/2016 | intravenous | humanized | PD-L1 | Urothelial carcinoma |
761034 | Link |
| atezolizumab | Tecentriq | Genentech | 10/18/2016 | intravenous | humanized | PD-L1 | non-small cell lung cancer |
761041 | Link |
| avelumab | Bavencio | EMD Serono |
3/23/2017 | intravenous | fully human | PD-L1 | Metastatic Merkel cell carcinoma |
761049 | Link |
| basiliximab | Simulect | Novartis | 5/12/1998 | intravenous | chimeric | IL2RA | Prophylaxis of renal transplant |
103764 | Link |
| belimumab | Benlysta | Human Genome Sciences | 3/9/2011 | intravenous | fully human | BLyS |
Systemic lupus erythematosus |
125370 | Link |
| bevacizumab | Avastin | Genentech | 2/26/2004 | intravenous | humanized | VEGF |
Metastatic colorectal cancer | 125085 | Link |
| bezlotoxumab | Zinplava | Merck | 10/21/2016 | intravenous | fully human | Clostridium difficile toxin B |
Prevent recurrence of Clostridium difficile infection |
761046 | Link |
| blinatumomab | Blincyto | Amgen | 12/3/2014 | intravenous | mouse, bispecific | CD19 | Precursor B-cell acute lymphoblastic leukemia |
125557 | Link |
| brentuximab vedotin | Adcetris | Seattle Genetics |
9/19/2011 | intravenous | chimeric, antibody-drug conjugate |
CD30 | Hodgkin lymphoma Anaplastic large-cell lymphoma |
125388 | Link |
| brodalumab | Siliq | Valeant |
2/15/2017 | subcutaneous | chimeric | IL17RA | Plaque psoriasis |
761032 | Link |
| canakinumab | Ilaris | Novartis | 6/17/2009 | subcutaneous | fully human | IL1B |
Cryopyrin-associated periodic syndrome | 125319 | Link |
capromab pendetide |
ProstaScint | Cytogen | 10/28/1996 | intravenous | murine, radiolabeled | PSMA | Diagnostic imaging agent in newly diagnosed prostate cancer or post-prostatectomy | 103608 | Link |
| certolizumab pegol | Cimzia | UCB (company) | 4/22/2008 | subcutaneous | humanized | TNF |
Crohn's disease | 125160 | Link |
| cetuximab | Erbitux | ImClone Systems | 2/12/2004 | intravenous | chimeric | EGFR | Metastatic colorectal carcinoma |
125084 | Link |
| daclizumab | Zenapax | Roche |
12/10/1997 | intravenous | humanized | IL2RA | Prophylaxis of renal transplant |
103749 | Link |
| daclizumab | Zinbryta | Biogen | 5/27/2016 | subcutaneous | humanized | IL2R |
Multiple sclerosis | 761029 | Link |
| daratumumab | Darzalex | Janssen Biotech |
11/16/2015 | intravenous | fully human | CD38 | Multiple myeloma | 761036 | Link |
| denosumab | Prolia, Xgeva | Amgen | 6/1/2010 | subcutaneous | fully human | RANKL | Postmenopausal women with osteoporosis |
125320 | Link |
| dinutuximab | Unituxin | United Therapeutics | 3/10/2015 | intravenous | chimeric | GD2 | Pediatric high-risk neuroblastoma | 125516 | Link |
| dupilumab | Dupixent | Regeneron Pharmaceuticals | 3/28/2017 | subcutaneous | fully human | IL4RA |
Atopic dermatitis, asthma | 761055 | Link |
| durvalumab | Imfinzi | AstraZeneca | 5/1/2017 | intravenous | fully human | PD-L1 | Urothelial carcinoma |
761069 | Link |
| eculizumab | Soliris | Alexion | 3/16/2007 | intravenous | humanized | Complement component 5 | Paroxysmal nocturnal hemoglobinuria | 125166 | Link |
| elotuzumab | Empliciti | Bristol-Myers Squibb |
11/30/2015 | intravenous | humanized | SLAMF7 | Multiple myeloma | 761035 | Link |
| evolocumab | Repatha | Amgen | 8/27/2015 | subcutaneous | fully human | PCSK9 | Heterozygous familial hypercholesterolemia Refractory hypercholesterolemia |
125522 | Link |
| golimumab | Simponi | Centocor |
4/24/2009 | subcutaneous | fully human | TNF |
Rheumatoid arthritis Psoriatic arthritis Ankylosing spondylitis |
125289 | Link |
| golimumab | Simponi Aria | Janssen Biotech |
7/18/2013 | intravenous | fully human | TNF |
Rheumatoid arthritis | 125433 | Link |
| ibritumomab tiuxetan | Zevalin | Spectrum Pharmaceuticals | 2/19/2002 | intravenous | murine, radioimmunotherapy | CD20 | Relapsed or refractory low-grade, follicular, or transformed B-cell non-Hodgkin's lymphoma |
125019 | Link |
| idarucizumab | Praxbind | Boehringer Ingelheim | 10/16/2015 | intravenous | humanized Fab | dabigatran | Emergency reversal of anticoagulant dabigatran | 761025 | Link |
| infliximab | Remicade | Centocor |
8/24/1998 | intravenous | chimeric | TNF alpha |
Crohn's disease | 103772 | Link |
infliximab-abda |
Renflexis | Samsung Bioepis |
4/21/2017 | intravenous | chimeric, biosimilar | TNF |
Plaque psoriasis |
761054 | Link |
infliximab-dyyb |
Inflectra | Celltrion Healthcare |
4/5/2016 | intravenous | chimeric, biosimilar | TNF |
Plaque psoriasis |
125544 | Link |
| ipilimumab | Yervoy | Bristol-Myers Squibb |
3/25/2011 | intravenous | fully human | CTLA-4 |
Metastatic melanoma |
125377 | Link |
| ixekizumab | Taltz | Eli Lilly | 3/22/2016 | subcutaneous | humanized | IL17A |
Plaque psoriasis |
125521 | Link |
| mepolizumab | Nucala | GlaxoSmithKline |
11/4/2015 | subcutaneous | humanized | IL5 | Severe asthma | 125526 | Link |
| natalizumab | Tysabri | Biogen Idec |
11/23/2004 | intravenous | humanized | alpha-4 integrin | Multiple sclerosis | 125104 | Link |
| necitumumab | Portrazza | Eli Lilly | 11/24/2015 | intravenous | fully human | EGFR | Metastatic squamous non-small cell lung carcinoma |
125547 | Link |
| nivolumab | Opdivo | Bristol-Myers Squibb |
12/22/2014 | intravenous | fully human | PD-1 |
Metastatic melanoma |
125554 | Link |
| nivolumab | Opdivo | Bristol-Myers Squibb |
3/4/2015 | intravenous | fully human | PD-1 |
Metastatic squamous non-small cell lung carcinoma |
125527 | Link |
| obiltoxaximab | Anthem | Elusys Therapeutics | 3/18/2016 | intravenous | chimeric | Protective antigen of the Anthrax toxin | Inhalational anthrax |
125509 | Link |
| obinutuzumab | Gazyva | Genentech | 11/1/2013 | intravenous | humanized | CD20 | Chronic lymphocytic leukemia | 125486 | Link |
| ocrelizumab | Ocrevus | Genentech | 3/28/2017 | intravenous | humanized | CD20 | Multiple sclerosis | 761053 | Link |
| ofatumumab | Arzerra | Glaxo Grp | 10/26/2009 | intravenous | fully human | CD20 | Chronic lymphocytic leukemia | 125326 | Link |
| olaratumab | Lartruvo | Eli Lilly | 10/19/2016 | intravenous | fully human | PDGFRA |
Soft tissue sarcoma |
761038 | Link |
| omalizumab | Xolair | Genentech | 6/20/2003 | subcutaneous | humanized | IgE |
Moderate to severe persistent asthma | 103976 | Link |
| palivizumab | Synagis | MedImmune | 6/19/1998 | intramuscular | humanized | F protein of RSV | Respiratory syncytial virus | 103770 | Link |
| panitumumab | Vectibix | Amgen | 9/27/2006 | intravenous | fully human | EGFR | Metastatic colorectal cancer | 125147 | Link |
| pembrolizumab | Keytruda | Merck | 9/4/2014 | intravenous | humanized | PD-1 |
Metastatic melanoma |
125514 | Link |
| pertuzumab | Perjeta | Genentech | 6/8/2012 | intravenous | humanized | HER2 | Metastatic breast cancer | 125409 | Link |
| ramucirumab | Cyramza | Eli Lilly | 4/21/2014 | intravenous | fully human | VEGFR2 |
Gastric cancer |
125477 | Link |
| ranibizumab | Lucentis | Genentech | 6/30/2006 | intravitreal injection | humanized | VEGFR2 |
Wet age-related macular degeneration |
125156 | Link |
| raxibacumab | Raxibacumab | Human Genome Sciences | 12/24/2012 | intravenous | fully human | Protective antigen of Bacillus anthracis | Inhalational anthrax |
125349 | Link |
| reslizumab | Cinqair | Teva |
3/23/2016 | intravenous | humanized | IL5 | Severe asthma | 761033 | Link |
| rituximab | Rituxan | Genentech | 11/26/1997 | intravenous | chimeric | CD20 | B-cell non-Hodgkin's lymphoma |
103705 | Link |
| secukinumab | Cosentyx | Novartis | 1/21/2015 | subcutaneous | fully human | IL17A |
Plaque psoriasis |
125504 | Link |
| siltuximab | Sylvant | Janssen Biotech |
4/23/2014 | intravenous | chimeric | IL6 | Multicentric Castleman's disease |
125496 | Link |
| tocilizumab | Actemra | Genentech | 1/8/2010 | intravenous | humanized | IL6R |
Rheumatoid arthritis | 125276 | Link |
| tocilizumab | Actemra | Genentech | 10/21/2013 | intravenous subcutaneous |
humanized | IL6R |
Rheumatoid arthritis Polyarticular juvenile idiopathic arthritis Systemic juvenile idiopathic arthritis |
125472 | Link |
| trastuzumab | Herceptin | Genentech | 9/25/1998 | intravenous | humanized | HER2 | Metastatic breast cancer | 103792 | Link |
| ustekinumab | Stelara | Centocor |
9/25/2009 | subcutaneous | fully human | IL12 IL23 |
Plaque psoriasis |
125261 | Link |
| ustekinumab | Stelara | Janssen Biotech |
9/23/2016 | subcutaneous intravenous |
fully human | IL12 IL23 |
761044 | Link | |
| vedolizumab | Entyvio | Takeda | 5/20/2014 | intravenous | humanized | integrin receptor | Ulcerative colitis Crohn's disease |
125476 | Link |
| sarilumab | Kevzara | Sanofi Aventis |
5/22/17 | subcutaneous | fully human | IL6R |
Rheumatoid arthritis | 761037 | Link |
| rituximab and hyaluronidase | Rituxan Hycela | Genentech | 6/22/17 | subcutaneous | chimeric, co-formulated | CD20 | Follicular lymphoma Diffuse large B-cell lymphoma Chronic lymphocytic leukemia |
761064 | Link |
| guselkumab | Tremfya | Janssen Biotech |
7/13/17 | subcutaneous | fully human | IL23 | Plaque psoriasis |
761061 | Link |
| inotuzumab ozogamicin | Besponsa | Wyeth | 8/17/17 | intravenous | humanized, antibody-drug conjugate |
CD22 | Precursor B-cell acute lymphoblastic leukemia |
761040 | Link |
adalimumab-adbm |
Cyltezo | Boehringer Ingelheim | 8/25/17 | subcutaneous | fully human, biosimilar | TNF |
Plaque psoriasis |
761058 | Link |
| gemtuzumab ozogamicin | Mylotarg | Wyeth | 9/1/17 | intravenous | humanized, antibody-drug conjugate |
CD33 | Acute myeloid leukemia | 761060 | Link |
bevacizumab-awwb |
Mvasi | Amgen | 9/14/17 | intravenous | humanized, biosimilar | VEGF |
Metastatic | 761028 | Link |
| benralizumab | Fasenra | AstraZeneca | 11/14/17 | subcutaneous | humanized | interleukin-5 receptor alpha subunit | Severe asthma, eosinophilic phenotype | 761070 | Link |
emicizumab-kxwh |
Hemlibra | Genentech | 11/16/17 | subcutaneous | humanized, bispecific | Factor IXa, Factor X | Hemophilia A (congenital Factor VIII deficiency) with Factor VIII inhibitors. |
761083 | Link |
trastuzumab-dkst |
Ogivri | Mylan | 12/1/17 | intravenous | humanized, biosimilar | HER2 | HER2-overexpressing breast cancer, metaststic gastric or gastroesophageal junction adenocarcinoma | 761074 | Link |
| infliximab-qbtx | Ixifi | Pfizer | 12/13/17 | intravenous | chimeric, biosimilar | TNF |
Plaque psoriasis |
761072 | Link |
ibalizumab-uiyk |
Trogarzo | TaiMed Biologics | 3/6/18 | intravenous | humanized | CD4 | HIV | 761065 | Link |
tildrakizumab-asmn |
Ilumya | Merck | 3/20/18 | subcutaneous | humanized | IL23 | Plaque psoriasis |
761067 | Link |
burosumab-twza |
Crysvita | Ultragenyx | 4/17/18 | subcutaneous | fully human | FGF23 |
X-linked hypophosphatemia | 761068 | Link |
erenumab-aooe |
Aimovig | Amgen | 5/17/18 | subcutaneous | fully human | CGRP receptor |
Migraine headache prevention |
761077 | Link |
Tositumomab – Bexxar – 2003 – CD20
Mogamulizumab – Poteligeo – August 2018 – CCR4
Moxetumomab pasudotox – Lumoxiti – September 2018 – CD22
Polatuzumab vedotin – Polivy – June 2019 – CD79B
The
Economics
Since 2000, the therapeutic market for monoclonal antibodies has grown exponentially. In 2006, the "big 5" therapeutic antibodies on the market were
References
- ^ PMID 23370250.
- ISBN 978-0-8153-4101-7.
- ^ ISBN 978-0-443-07310-6.
- ^ PMID 18045976.
- S2CID 10169378.
- S2CID 43781004.
- S2CID 4161444.
- PMID 7427932.
- PMID 7032624.
- ^ PMID 15780905.
- ^ S2CID 19243664.
- PMID 1350088.
- S2CID 904440.
- S2CID 4241051.
- S2CID 9745527.
- PMID 20073133.
- PMID 20065651.
- S2CID 21298489.
- PMID 26784573.
- ^ S2CID 4608450.
- PMID 28335888.
- ^ ISBN 978-0-443-07145-4.
- ^ S2CID 19375883.
- ^ PMID 28967385.
- ^ PMID 38219962.
- ^ S2CID 26323381.
- PMID 28967385.
- ^ Goel, Ayush (20 August 2013). "Vasogenic cerebral oedema". radiopaedia.org. Retrieved 2017-11-01.
- ^ PMID 21091109.
- PMID 37458272.
- PMID 27048170.
- S2CID 242999976.
- PMID 36449413.
- ^ "Leqembi | ALZFORUM". www.alzforum.org. Retrieved 2024-02-14.
- ^ Commissioner, Office of the (2023-07-07). "FDA Converts Novel Alzheimer's Disease Treatment to Traditional Approval". FDA. Retrieved 2024-02-14.
- PMID 12237768.
- PMID 15687597.
- S2CID 11603692.
- S2CID 25271222.
- PMID 20073127.
- PMID 20190561.
- PMID 20061824.
- PMID 20065641.
