Homologation reaction
In
methoxymethylenetriphenylphosphine
to give the next homologue in the series.
Examples of homologation reactions include:
- Kiliani-Fischer synthesis, where an aldosemolecule is elongated through a three-step process consisting of:
- Nucleophillic addition of cyanide to the carbonyl to form a cyanohydrin
- Hydrolysis to form a lactone
- Reduction to form the homologous aldose
- methoxymethylenetriphenylphosphine, which produces a homologous aldehyde.
- Arndt–Eistert reaction is a series of chemical reactions designed to convert a carboxylic acid to a higher carboxylic acid homologue (i.e. contains one additional carbon atom)
- ynolate intermediate.[2]
- Seyferth–Gilbert homologation in which an aldehyde is converted to a terminal alkyne and then hydrolyzed back to an aldehyde.
Some reactions increase the chain length by more than one unit. For example, the DeMayo reaction can be considered a two-carbon homologation reaction.
Chain reduction
Likewise the chain length can also be reduced:
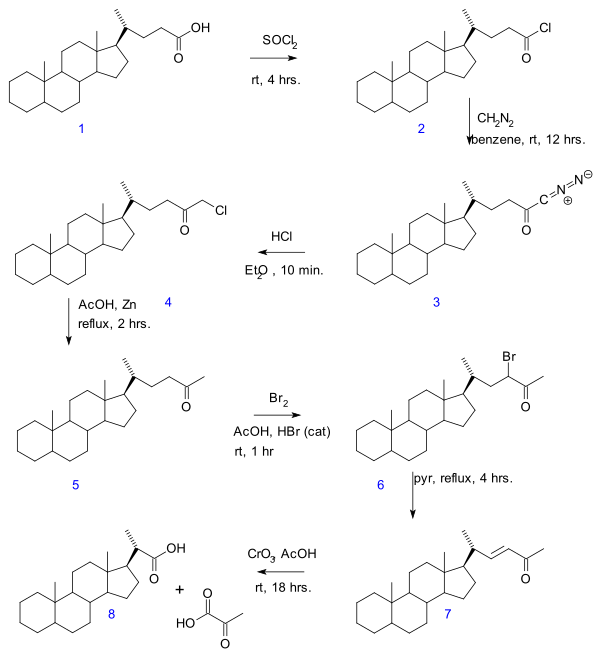
- In the enone 7 and finally oxidation with chromium trioxideto bisnorcholanic acid 8.
- In the

- Mechanistically oxidation causes ring-cleavage at the alkene group, extrusion of carbon dioxide in decarboxylation with subsequent ring-closure.