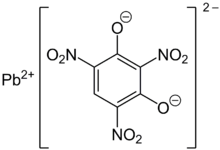
Lead styphnate
| |
| Names | |
|---|---|
| Preferred IUPAC name
Lead(II) 2,4,6-trinitrobenzene-1,3-bis(olate) | |
| Other names
Lead 2,4,6-trinitrobenzene-1,3-diolate
Lead 2,4,6-trinitro-m-phenylene dioxide 1,3-Benzenediol, 2,4,6-trinitro-, lead(2+) salt (1:1) Lead tricinate Lead trinitroresorcinate Tricinat[1] | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.035.703 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 0130 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6HN3O8Pb | |
| Molar mass | 450.288 g/mol |
| Density | 3.06 to 3.1 g cm−3 |
| Explosive data | |
| Shock sensitivity | High |
| Friction sensitivity | High |
| Detonation velocity | 5200 m/s |
| Hazards | |
| GHS labelling:[2] | |
   
| |
| Danger | |
| H200, H302, H332, H360Df, H373, H410 | |
| NFPA 704 (fire diamond) | |
| 330 °C (626 °F; 603 K) | |
| Safety data sheet (SDS) | Oxford MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lead styphnate (lead 2,4,6-
Lead styphnate forms six-sided crystals of the
Preparation
Although never substantiated, lead styphnate may have been discovered by Peter Griess (of Griess test fame) in 1874. In 1919, Edmund Herz first established a preparation of anhydrous normal lead styphnate by the reaction of magnesium styphnate with lead acetate in the presence of nitric acid.[4][3]
- {C6N3O8}MgH2O + Pb(CH3CO2)2 → {C6N3O8}PbH2O + Mg(CH3CO2)2
Structure
Normal lead styphnate exists as α and β polymorphs, both being monoclinic crystals. The lead centres are seven-coordinate and are bridged via oxygen bridges. The water molecule is coordinated to the metal and is also hydrogen-bonded to the anion. Many of the Pb-O distances are short, indicating some degree of covalency. The styphnate ions lie in approximately parallel planes linked by Pb atoms.[5][6]
Properties
Lead styphnate's heat of formation is −835 kJ mol−1. The loss of water leads to the formation of a sensitive anhydrous material with a density of 2.9 g cm−3. The variation of colors remains unexplained.[7] Lead styphnate has a detonation velocity of 5.2 km/s and an explosion temperature of 265–280 °C after five seconds.[8]
Applications
Lead styphnate is mainly used in small arms
References
- ^ ECHA, European Chemicals Agency "Archived copy" (PDF). Archived from the original (PDF) on 2014-10-22. Retrieved 2014-10-17.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ GHS: GESTIS 490561
- ^ ISBN 978-3527306732
- .
- .
- .
- S2CID 199492549.
- .
- ^ Gray, Theodore (2009). "Flash Bang". Popular Science.
- ^ Daniel W. Youngner; et al. (2000). "MEMS Mega-pixel Micro-thruster Arrays for Small Satellite Stationkeeping". Honeywell Technology 14th Annual/USU Conference on Small Satellites. Archived from the original on 2021-03-10. Retrieved 2016-10-18.

