Lead(II) acetate

| |

| |
| Names | |
|---|---|
| IUPAC name
Lead(II) acetate
| |
| Systematic IUPAC name
Lead(II) ethanoate | |
| Other names
Plumbous acetate, Salt of Saturn, Sugar of Lead, Lead diacetate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.005.551 |
| EC Number |
|
| MeSH | lead+acetate |
PubChem CID
|
|
RTECS number
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Pb(C2H3O2)2 | |
| Molar mass | 325.29 g/mol (anhydrous) 379.33g/mol (trihydrate) |
| Appearance | White powder or colourless, efflorescent crystals |
| Odor | Slightly acetic |
| Density | 3.25 g/cm3 (20 °C, anhydrous) 2.55 g/cm3 (trihydrate) 1.69 g/cm3 (decahydrate)[1] |
| Melting point | 280 °C (536 °F; 553 K) (anhydrous) 75 °C (167 °F; 348 K) (trihydrate) decomposes[4] at ≥ 200 °C 22 °C (72 °F; 295 K) (decahydrate)[1] |
| Boiling point | Decomposes |
| Anhydrous: 19.8 g/100 mL (0 °C) 44.31 g/100 mL (20 °C) 69.5 g/100 mL (30 °C)[2] 218.3 g/100 mL (50 °C)[1] | |
| Solubility | Anhydrous and trihydrate are soluble in alcohol, glycerol[2] |
| Solubility in methanol | Anhydrous:[2] 102.75 g/100 g (66.1 °C) Trihydrate:[3] 74.75 g/100 g (15 °C) 214.95 g/100 g (66.1 °C) |
| Solubility in glycerol | Anhydrous:[2] 20 g/100 g (15 °C) Trihydrate:[3] 143 g/100 g (20 °C) |
| −89.1·10−6 cm3/mol | |
Refractive index (nD)
|
1.567 (trihydrate)[1] |
| Structure | |
| Monoclinic (anhydrous, trihydrate) Rhombic (decahydrate) | |
| Thermochemistry | |
Std enthalpy of (ΔfH⦵298)formation |
−960.9 kJ/mol (anhydrous)[2] −1848.6 kJ/mol (trihydrate)[3] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Neurotoxic, probable human carcinogen
|
| GHS labelling: | |
  [4] [4]
| |
| Danger | |
| H360, H373, H410[4] | |
| P201, P273, P308+P313, P501[4] | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
400 mg/kg (mice, oral)[1] |
LCLo (lowest published)
|
300 mg/kg (dog, oral)[5] |
| Related compounds | |
Other cations
|
Lead(IV) acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lead(II) acetate, also known as lead acetate, lead diacetate, plumbous acetate, sugar of lead, lead sugar, salt of
The substance is used as a reagent to make other lead compounds and as a fixative for some dyes. In low concentrations, it formerly served as the principal active ingredient in progressive types of
Production
Lead(II) acetate can be made by boiling elemental lead in acetic acid and hydrogen peroxide. This method will also work with
- Pb + H2O2 + 2 H+ → Pb2+ + 2 H2O
- Pb2+ + 2 AcO− → Pb(OAc)2
Lead(II) acetate can also be made by dissolving lead(II) oxide in acetic acid:[7]
- PbO + 2 AcOH → Pb(OAc)2 + H2O
Lead(II) acetate can also be made via a
- Cu(OAc)2 + Pb → Cu + Pb(OAc)2
Structure
The crystal structure of anhydrous lead(II) acetate has been described as a 2D coordination polymer. In comparison, lead(II) acetate trihydrate's structure is a 1D coordination polymer.[8] In the trihydrate, the Pb2+ ion's coordination sphere consists of nine oxygen atoms belonging to three water molecules, two bidentate acetate groups and two bridging acetate groups. The coordination geometry at Pb is a monocapped square antiprism.[9][10] The trihydrate thermally decomposes to a hemihydrate, Pb(OAc)2·1⁄2H2O, and to basic acetates such as Pb4O(OAc)6 and Pb2O(OAc)2.[8]
| Anhydrous[8] Pb(OAc)2 |
Trihydrate[9][10] Pb(OAc)2·3H2O | |
|---|---|---|
| Lead coordination sphere |

|

|
| Strongly bonded aggregation |
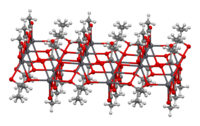 2D sheet |
 1D chain |
| Weakly bonded aggregation |
hydrophobic surfaces in contact
|
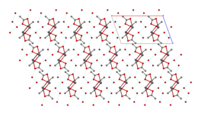 chains linked by hydrogen bonds |
Uses
Sweetener
Like other lead(II) salts, lead(II) acetate has a sweet taste, which led to its historical use as a sugar substitute in both wines and foods.[11] The
Historical incidents
The earliest confirmed poisoning by lead acetate was that of Pope Clement II who died in October 1047. A toxicological examination of his remains conducted in the mid-20th century confirmed centuries-old rumors that he had been poisoned with lead sugar.[14] It is not clear whether he was assassinated.
In 1787 painter and biographer Albert Christoph Dies swallowed, by accident, approximately 3/4 oz (20 g) of lead acetate. His recovery from this poison was slow and incomplete. He lived with illnesses until his death in 1822.[15][16]
Although the use of lead(II) acetate as a sweetener was already illegal at that time, composer
In the 1850s, Mary Seacole applied lead(II) acetate, among other remedies, against an epidemic of cholera in Panama.[19][20]
In 1887, 38 hunting horses belonging to Captain William Hollwey Steeds were poisoned in their stables at Clonsilla House, Dublin, Ireland. At least ten of the hunters died. Captain Steeds, an "extensive commission agent", had previously supplied the horses for the Bray and Greystones Coach. It transpired that they had been fed a bran mash that had been sweetened with a toxic lead acetate.[21]
Cosmetics
Lead(II) acetate, as well as white lead, has been used in cosmetics throughout history.[22]
Until recently,[
Medical uses
Lead(II) acetate solution was a commonly used folk remedy for sore nipples.
Industrial uses
Lead(II) acetate paper is used to detect the poisonous gas hydrogen sulfide. The gas reacts with lead(II) acetate on the moistened test paper to form a grey precipitate of lead(II) sulfide.
An aqueous solution of lead(II) acetate is the byproduct of a 1:1 ratio of
Other uses
It was also used in making of slow matches during the Middle Ages. It was made by mixing a natural form of lead(II) oxide called litharge and vinegar.
Sugar of lead was a recommended agent added to linseed oil during heating to produce "boiled" linseed oil, the lead and heat acting to cause the oil to cure faster than raw linseed oil.[28]
Lead(II) acetate ("salt of Saturn") was used to synthesise acetone which was then known as "spirit of Saturn" for being made with the salt of Saturn and thought to be a lead compound in the 17th century.[29]
See also
- Saturn's Tree
References
- ^ ISBN 0-07-049439-8.
- ^ a b c d e "Lead(II) acetate".
- ^ a b c "Lead(II) acetate trihydrate".
- ^ a b c d Sigma-Aldrich Co., Lead(II) acetate trihydrate. Retrieved on 2014-06-08.
- ^ "Lead compounds (as Pb)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "Lead Acetate in 'Progressive' Hair Dye Products". fda.gov. 7 October 2021.
- ISBN 978-0-08-037941-8.
- ^ PMID 27548299.
- ^ S2CID 201671682.
- ^ .
- ^ "The Disturbingly Long History of Lead Toxicity in Winemaking." Anna Archibald, 30 July 2020. Retrieved: 22 December, 2020.
- ^ Lead Poisoning and Rome
- ISBN 9780080875606,
From the results achieved so far it is obvious that the purity law for lead in wines in the last two centuries was frequently ignored.
- ^ Specht, W. and Fischer, K. (1959). "Vergiftungsnachweis an den Resten einer 900 Jahre alten Leiche" [Evidence of poisoning in the remains of a 900-year-old corpse] (in German). Archiv für Kriminologie 124: 61–84.
- ^ One or more of the preceding sentences incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Dies, Christoph Albert". Encyclopædia Britannica. Vol. 8 (11th ed.). Cambridge University Press. p. 211.
- ISBN 0-299-02791-0.
- ^ "Beethoven und Blei: Tödliches Zusammenspiel". Archived from the original on 2009-02-21. Retrieved 2009-09-12.
- ^ "Beethoven litt unter Bleivergiftung". Archived from the original on 2009-02-21. Retrieved 2020-02-24.
- ISBN 0-14-043902-1
- ISBN 1-84119-677-0
- ^ Weekly Irish Times, Saturday 15 October 1887; Enniskillen Chronicle and Erne Packet, 24 October 1887, p. 3
- ^ Gunn, Fenja. (1973) The Artificial Face: A History of Cosmetics. — as cited in Leisure Activities of an 18th Century Lady
- ^ Lead Based Hair Products: Too Hazardous for Household Use - Results, Howard W. Mielke, PhD, Myiesha D. Taylor, Chris R. Gonzales, M. Kelley Smith, Pamela V. Daniels, and Ayanna V.Buckner. Journal of American Pharmaceutical Association (NS37, Jan/Feb 1997:85-89).
- ^ a b "Can West News Service: Grecian Formula in a grey zone after ban". Archived from the original on 2007-08-08.
- ^ "The Proposition 65 List". Archived (PDF) from the original on 2014-10-31. Retrieved 2014-11-01.
- ^ The American Frugal Housewife, by Lydia M. Child
- ^ Laboratory manual in biology. Sharpe. 1911, American Book Company. p. 351
- ^ Andés, Louis Edgar, and Arthur Morris. Oil colours and printers' inks a practical handbook treating of linseed oil, boiled oil, paints, artists' colours, lampblack and printers' inks, black and coloured. London: Scott, Greenwood. 1903. 41. Print.
- ^ Mel Gorman, History of acetone (1600-1850), 1962
External links
- Case Studies in Environmental Medicine - Lead Toxicity
- Essay on "Lead Poisoning and Rome"
- HowStuffWorks "What Kind of Hair Color Do Men Use?" discussion of progressive dyes containing lead acetate
- National Pollutant Inventory - Lead and Lead Compounds Fact sheet (Does Not Bring Up Lead)
- ToxFAQs: Lead
- US Food and Drug Administration (FDA) fact sheet "Lead Acetate in Hair Dye Products"
- US Food and Drug Administration (FDA)21CFR73.2396 "PART 73 -- LISTING OF COLOR ADDITIVES EXEMPT FROM CERTIFICATION, Subpart C--Cosmetics, Sec. 73.2396 Lead acetate"

