Mercury telluride
| |
| Names | |
|---|---|
| Systematic IUPAC name
Mercury telluride | |
| Other names
Mercuric telluride, mercury(II) telluride
| |
| Identifiers | |
3D model (
JSmol ) |
|
ECHA InfoCard
|
100.031.905 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| HgTe | |
| Molar mass | 328.19 g/mol |
| Appearance | near black cubic crystals |
| Density | 8.1 g/cm3 |
| Melting point | 670°C |
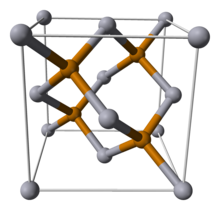
| Structure | |
| Sphalerite, cF8 | |
| F43m, No. 216 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mercury telluride (HgTe) is a binary chemical compound of
HgTe occurs in nature as the mineral form coloradoite.
Physical properties
All properties are at standard temperature and pressure unless stated otherwise. The lattice parameter is about 0.646 nm in the cubic crystalline form. The bulk modulus is about 42.1 GPa. The thermal expansion coefficient is about 5.2×10−6/K. Static dielectric constant 20.8, dynamic dielectric constant 15.1.
Doping
N-type doping can be achieved with elements such as boron, aluminium, gallium, or indium. Iodine and iron will also dope n-type. HgTe is naturally p-type due to mercury vacancies. P-type doping is also achieved by introducing zinc, copper, silver, or gold.[1][2]
Topological insulation

Mercury telluride was the first
Chemistry
HgTe bonds are weak. Their
Growth
Bulk growth is from a mercury and tellurium melt in the presence of a high mercury vapour pressure. HgTe can also be grown epitaxially, for example, by sputtering or by
Nanoparticles of mercury telluride can be obtained via cation exchange from cadmium telluride nanoplatelets.[6]
See also
References
External links
- Thermophysical properties database[permanent dead link] at Germany's Chemistry Information Centre, Berlin
