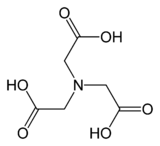
Nitrilotriacetic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,2′,2′′-Nitrilotriacetic acid[3] | |
| Other names | |
| Identifiers | |
3D model (
JSmol ) |
|
| 1710776 | |
| ChEBI | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.004.869 |
| EC Number |
|
| 3726 | |
| KEGG | |
| MeSH | Nitrilotriacetic+Acid |
PubChem CID
|
|
RTECS number
|
|
| UNII |
|
| UN number | 2811 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H9NO6 | |
| Molar mass | 191.14 [4] |
| Appearance | White crystals |
| Melting point | 246[4] °C (475 °F; 519 K) |
| Insoluble. <0.01 g/100 mL at 23°C [4] | |
| Thermochemistry | |
Std enthalpy of (ΔfH⦵298)formation |
−1.3130–−1.3108 MJ mol−1 |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H302, H319, H351 | |
| P281, P305+P351+P338 | |
| Flash point | 100 °C (212 °F; 373 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
1.1 g kg−1 (oral, rat) |
| Related compounds | |
Related alkanoic acids
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nitrilotriacetic acid (NTA) is the
Production and use
Nitrilotriacetic acid is commercially available as the free acid and as the sodium salt. It is produced from
Coordination chemistry and applications
The conjugate base of NTA is a
Like
In one application, sodium NTA removes Cr, Cu, and As from wood that had been treated with chromated copper arsenate.[9]
Laboratory uses
In the laboratory, this compound is used in complexometric titrations. A variant of NTA is used for protein isolation and purification in the
The his-tag binds the metal of metal chelator complexes. Previously, iminodiacetic acid was used for that purpose. Now, nitrilotriacetic acid is more commonly used.[12]
For laboratory uses Ernst Hochuli et al. 1987 coupled the NTA ligand and Nickel-ions to agarose beads.[13] This Ni-NTA Agarose is the most used tool to purify his tagged proteins via affinity chromatography.
- NTA complexes
-
Three views of the structure of [Ni(NTA)(H2O)2]−.
-
Structure of the nitrilotriacetate anion [Ca(NTA)(H2O)3]−.
Toxicity and environment
In contrast to EDTA, NTA is easily biodegradable and is almost completely removed during wastewater treatment.[6] The environmental impacts of NTA are minimal. Despite widespread use in cleaning products, the concentration in the water supply is too low to have a sizeable impact on human health or environmental quality.[14]
Related compounds
- N-Methyliminodiacetic acid (MIDA), the N-methyl derivative of IDA
- Imidodiacetic acid, the amino diacetic acid
- N-(2-Carboxyethyl)iminodiacetic acid, a more biodegradable analogue of NTA
- N-hydroxyiminodiacetic acid (HIDA), HON(CH2CO2H)2 (HIDA scan.
References
- ^ "Nitrilotriacetic Acid - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification. Retrieved 13 July 2012.
- ^ Nitrilotriacetic acid
- ISBN 978-0-85404-182-4.
- ^ a b c ChemBK Chemical Database http://www.chembk.com/en/chem/Nitrilotriacetic%20acid
- ^ Nitrilotriacetic Acid and Its Salts, International Agency for Research on Cancer
- ^ ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - .
- ^ Fang-Chih, C.; Ya-Nang, W.; Pin-Jui, C.; Chun-Han, K. Factors affecting chelating extraction of Cr, Cu, and As from CCA-treated wood. J. Environ. Manag. 2013, 122.
- PMID 27042860.
- ^ qiaexpressionist
- PMID 12116359.
- PMID 3443622.
- ^ Brouwer, N.; Terpstra, P. Ecological and Toxicological Properties of Nitrilotriacetic Acid (NTA) as a Detergent Builder. Tenside Surfactants Detergents 1995, 32, 225-228.
- .

![Three views of the structure of [Ni(NTA)(H2O)2]−.](http://upload.wikimedia.org/wikipedia/commons/thumb/1/14/Ni%28NTA%29%28aq%2923views.png/500px-Ni%28NTA%29%28aq%2923views.png)
![Structure of the nitrilotriacetate anion [Ca(NTA)(H2O)3]−.](http://upload.wikimedia.org/wikipedia/commons/thumb/0/06/Calcium_complex_of_NTA_trianion.svg/210px-Calcium_complex_of_NTA_trianion.svg.png)