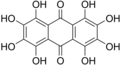
Octahydroxyanthraquinone
Appearance
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,2,3,4,5,6,7,8-Octahydroxyanthracene-9,10-dione | |||
| Other names
Octahydroxyanthracenedione
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C14H8O10 | |||
| Molar mass | 336.208 g·mol−1 | ||
| log P | -0.291 | ||
| Acidity (pKa) | 5.358 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Octahydroxyanthraquinone is an
hydroxyl
groups.
The compound was obtained in 1911 by
mercuric oxide in sulfuric acid at 250 °C (482 °F).[3]
LCD applications.[3]
Octahydroxyanthraquinone is active against the malaria parasite, but rufigallol (1,2,3,5,6,7-hexahydroxyanthraquinone) is 22 times more potent.[4]
References
- S2CID 97848374.
- ^ Wahl, Andre; Atack, F. W (1919) The Manufacture Of Organic Dyestuffs. G. Bell And Sons, Limited. Online version accessed on 2010-01-22.
- ^ S2CID 59445187.
- .